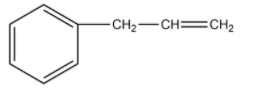
On oxymercuration-demercuration, the given compound produces the major product:
(A) 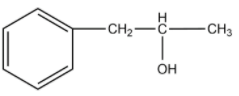
(B) 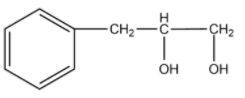
(C) 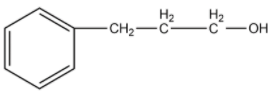
(D) 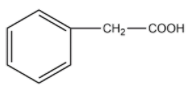
Answer
246k+ views
Hint: Recollect what happens in the oxymercuration-demercuration reaction. Identify which is the functional group of the reactant given in the question. Write the reaction equation and then determine which will be the most stable product to get the answer.
Complete step by step solution:
-Oxymercuration-demercuration is a two-step addition reaction which converts alkene to alcohol.
-In oxymercuration reaction, alkene reacts with mercuric acetate, ${{\left( C{{H}_{3}}COO \right)}_{2}}Hg$ in aqueous solution, which leads to addition of mercurous acetate$\left( C{{H}_{3}}COOHg \right)$ group and a hydroxyl group, -OH across the double bond.
-In the Demercuration reaction, the mercurous acetate$\left( C{{H}_{3}}COOHg \right)$ group will be replaced by hydrogen to form alkane.
-General reaction is represented as follows,
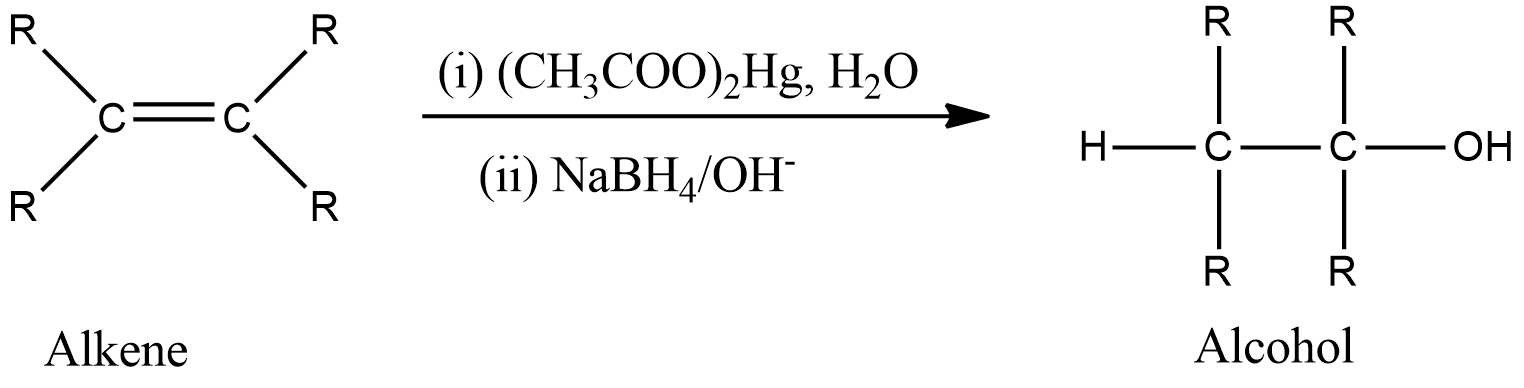
-The mechanism is shown below. First, there is the addition of mercuric acetate across the double bond to form a tricyclic intermediate which on hydrolysis forms an intermediate containing mercurous acetate group and alcohol group. Then, in the presence of reducing agents, demercuration occurs which removes mercurous acetate and reduces the intermediate to form the product that is alcohol.
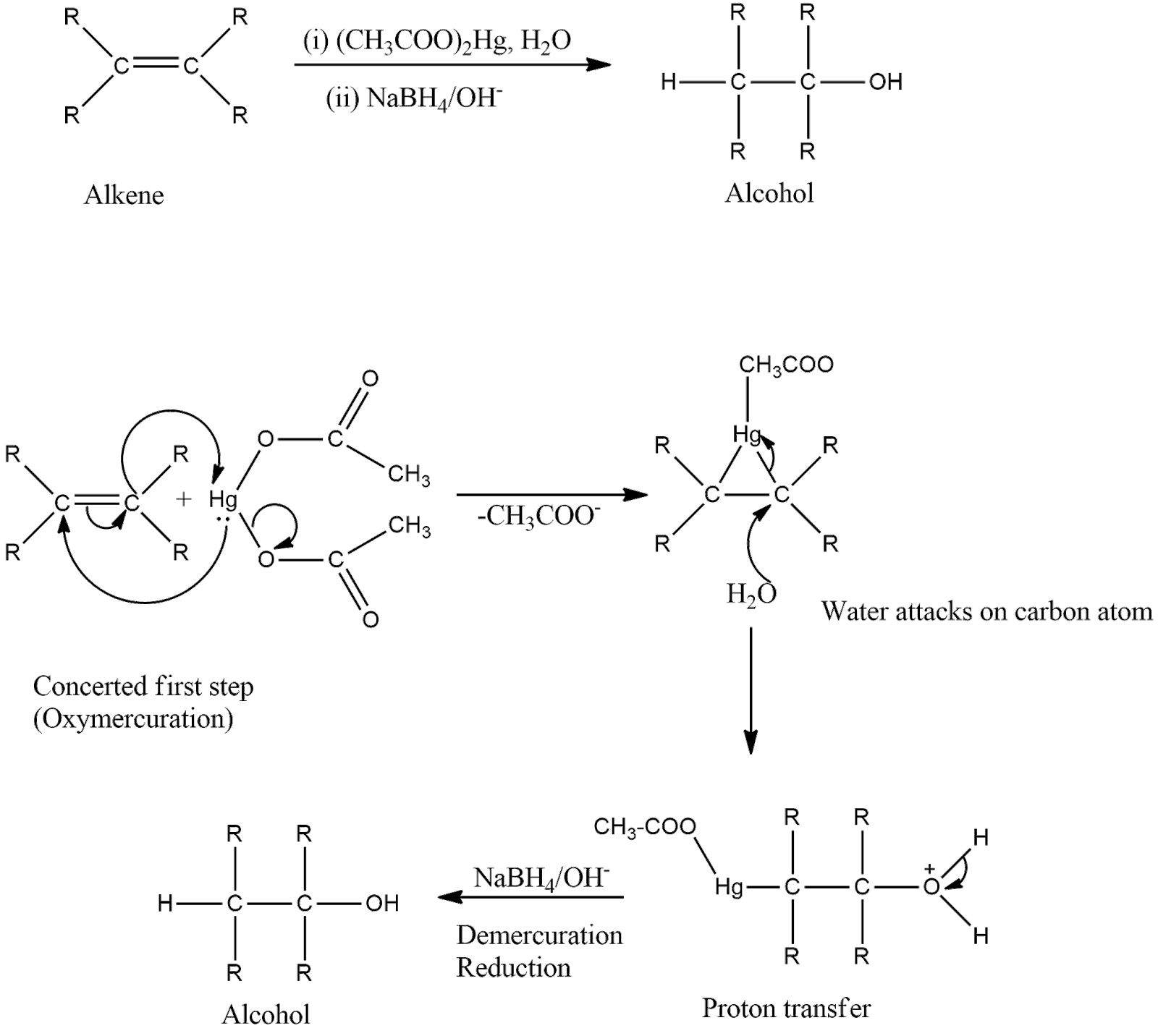
-In oxymercuration-demercuration reaction, carbocations are not formed, so rearrangements are not seen in this reaction.
-The reaction follows Markovnikoff's rule which states that the nucleophile will get added to that carbon atom in the double bond which has less number of hydrogen atoms and more number or substituents attached to it.
-The compound given as reactant is 3-phenylprop-1-ene. So, 3-phenylprop-1-ene will undergo oxymercuration-demercuration reaction to form 3-phenylpropan-2-ol and 3-phenylpropan-1-ol.
-But this reaction follows Markovnikoff’s rule and so, 3-phenylpropan-2-ol will be the major product.
-The reaction is represented as follows,
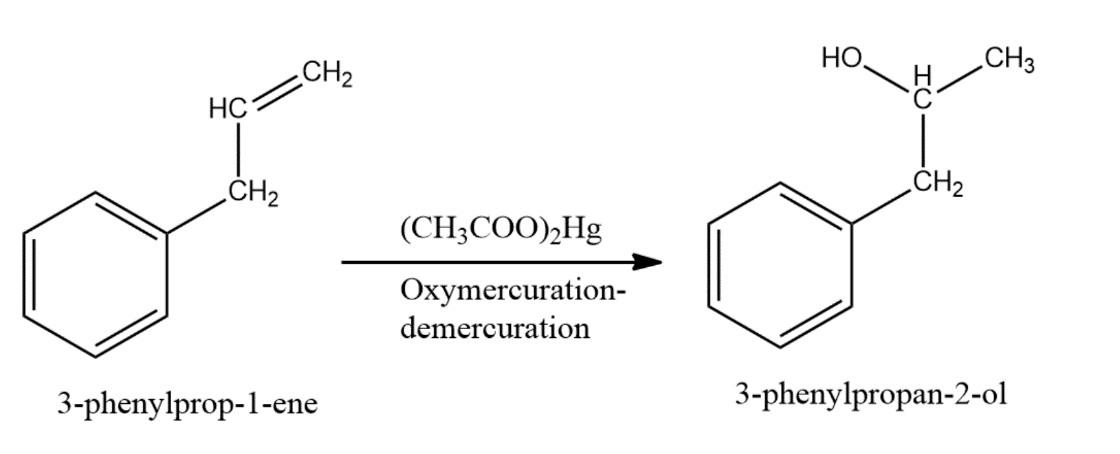
-Therefore, the given compound 3-phenylprop-1-ene on oxymercuration-demercuration reaction gives 3-phenylpropan-2-ol as the major product.
Therefore, the answer is option (A).
Note: In addition reactions of mixed unsaturated compounds, Markovnikoff’s rule is followed which tells us about the major product form in the reaction. Oxymercuration-demercuration reaction is an anti-addiction reaction and converts alkenes to neutral alcohols.
Complete step by step solution:
-Oxymercuration-demercuration is a two-step addition reaction which converts alkene to alcohol.
-In oxymercuration reaction, alkene reacts with mercuric acetate, ${{\left( C{{H}_{3}}COO \right)}_{2}}Hg$ in aqueous solution, which leads to addition of mercurous acetate$\left( C{{H}_{3}}COOHg \right)$ group and a hydroxyl group, -OH across the double bond.
-In the Demercuration reaction, the mercurous acetate$\left( C{{H}_{3}}COOHg \right)$ group will be replaced by hydrogen to form alkane.
-General reaction is represented as follows,
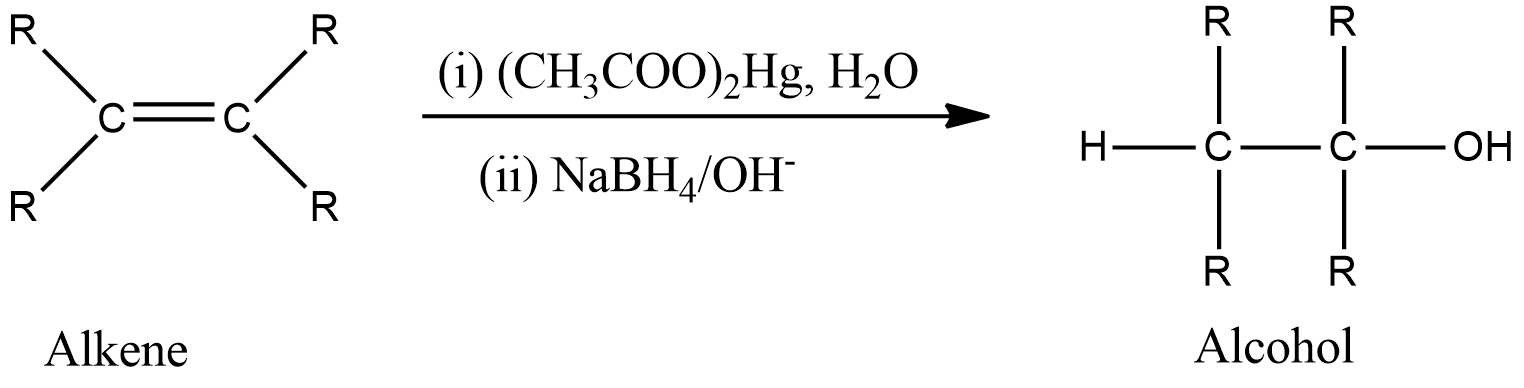
-The mechanism is shown below. First, there is the addition of mercuric acetate across the double bond to form a tricyclic intermediate which on hydrolysis forms an intermediate containing mercurous acetate group and alcohol group. Then, in the presence of reducing agents, demercuration occurs which removes mercurous acetate and reduces the intermediate to form the product that is alcohol.

-In oxymercuration-demercuration reaction, carbocations are not formed, so rearrangements are not seen in this reaction.
-The reaction follows Markovnikoff's rule which states that the nucleophile will get added to that carbon atom in the double bond which has less number of hydrogen atoms and more number or substituents attached to it.
-The compound given as reactant is 3-phenylprop-1-ene. So, 3-phenylprop-1-ene will undergo oxymercuration-demercuration reaction to form 3-phenylpropan-2-ol and 3-phenylpropan-1-ol.
-But this reaction follows Markovnikoff’s rule and so, 3-phenylpropan-2-ol will be the major product.
-The reaction is represented as follows,

-Therefore, the given compound 3-phenylprop-1-ene on oxymercuration-demercuration reaction gives 3-phenylpropan-2-ol as the major product.
Therefore, the answer is option (A).
Note: In addition reactions of mixed unsaturated compounds, Markovnikoff’s rule is followed which tells us about the major product form in the reaction. Oxymercuration-demercuration reaction is an anti-addiction reaction and converts alkenes to neutral alcohols.
Recently Updated Pages
Which of the following contains the highest percentage class 12 chemistry JEE_Main

Protein can be most easily removed from A Alkanes B class 12 chemistry JEE_Main

PhCOCHBr2xrightarrowOHAxrightarrowOHBxrightarrowH+C class 12 chemistry JEE_Main

When alcohol reacts with conc H2SO4 intermediate compound class 12 chemistry JEE_Main

A nanopeptide contains peptide linkages A 10 B 8 C class 12 chemistry JEE_Main

Which one of the following is paramagnetic A NO B N2O class 12 chemistry JEE_Main

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Collisions: Types and Examples for Students

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




