Which of the following shows optical activity?
A.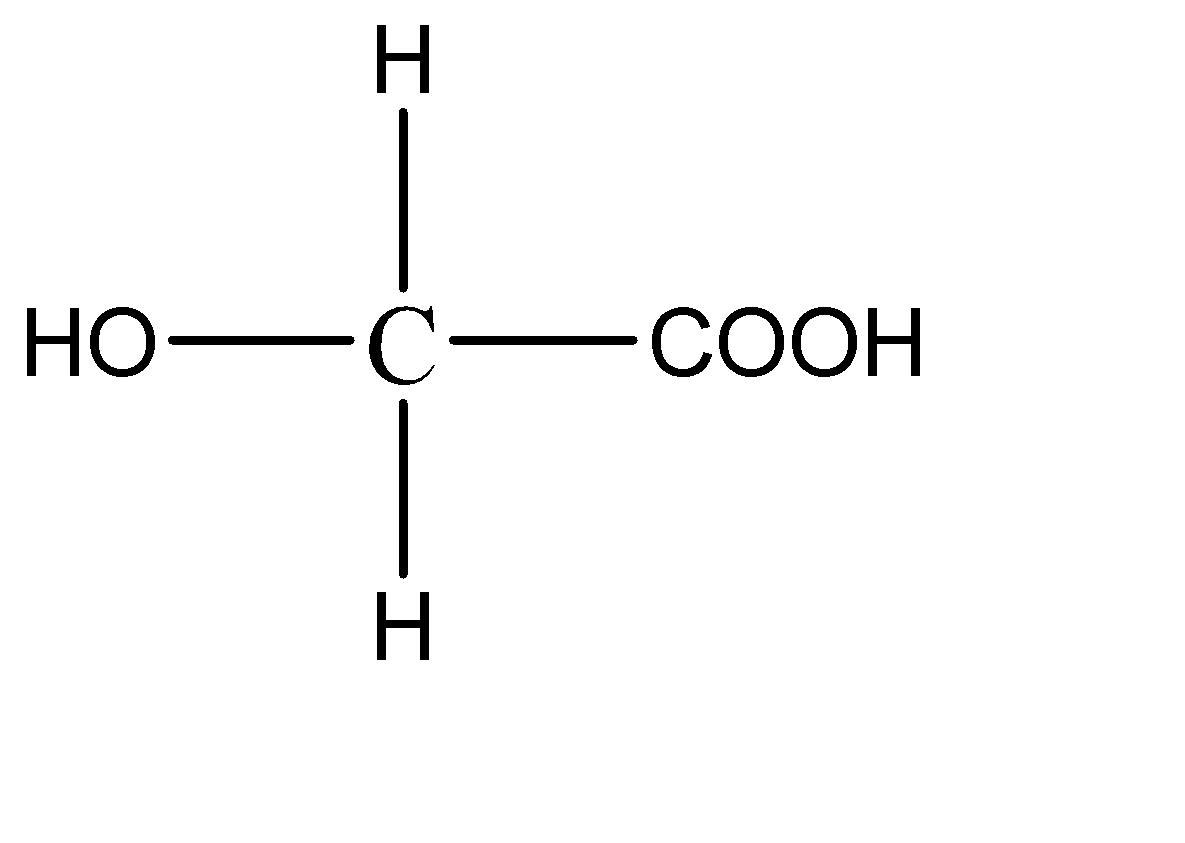
B.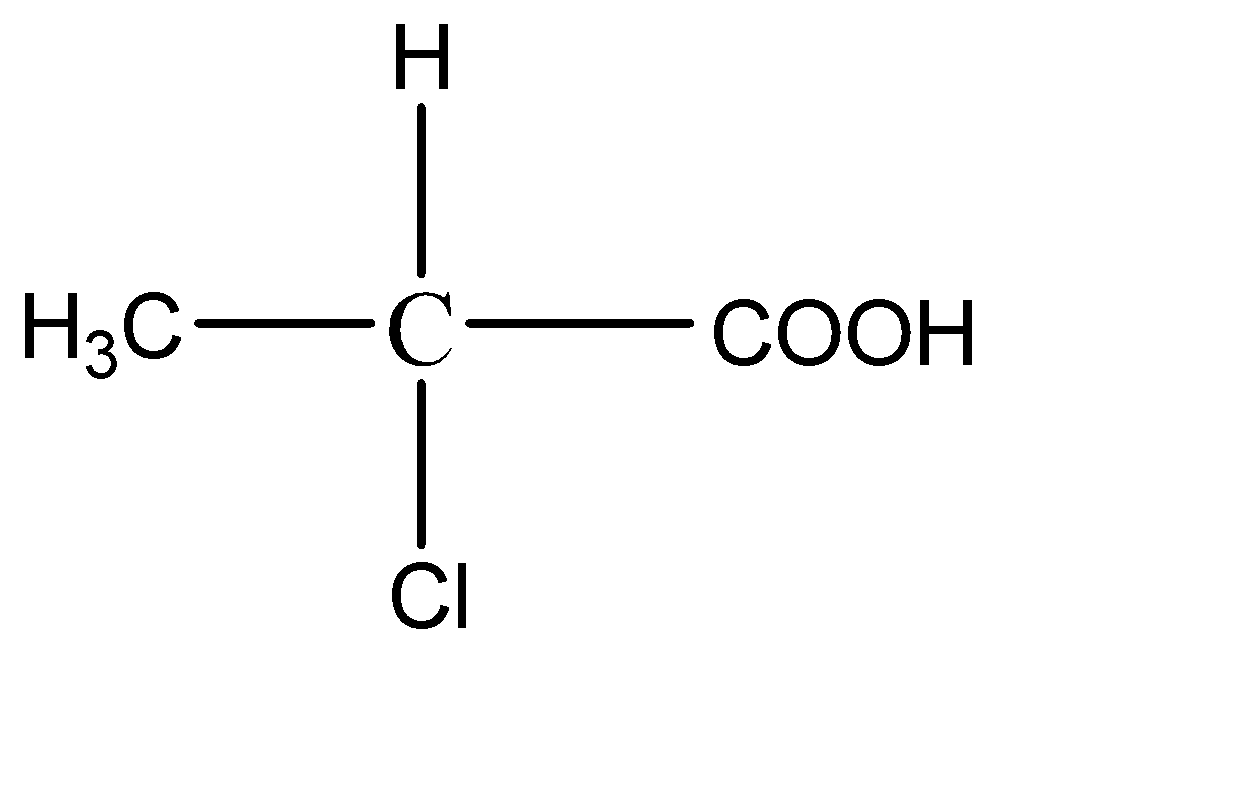
C.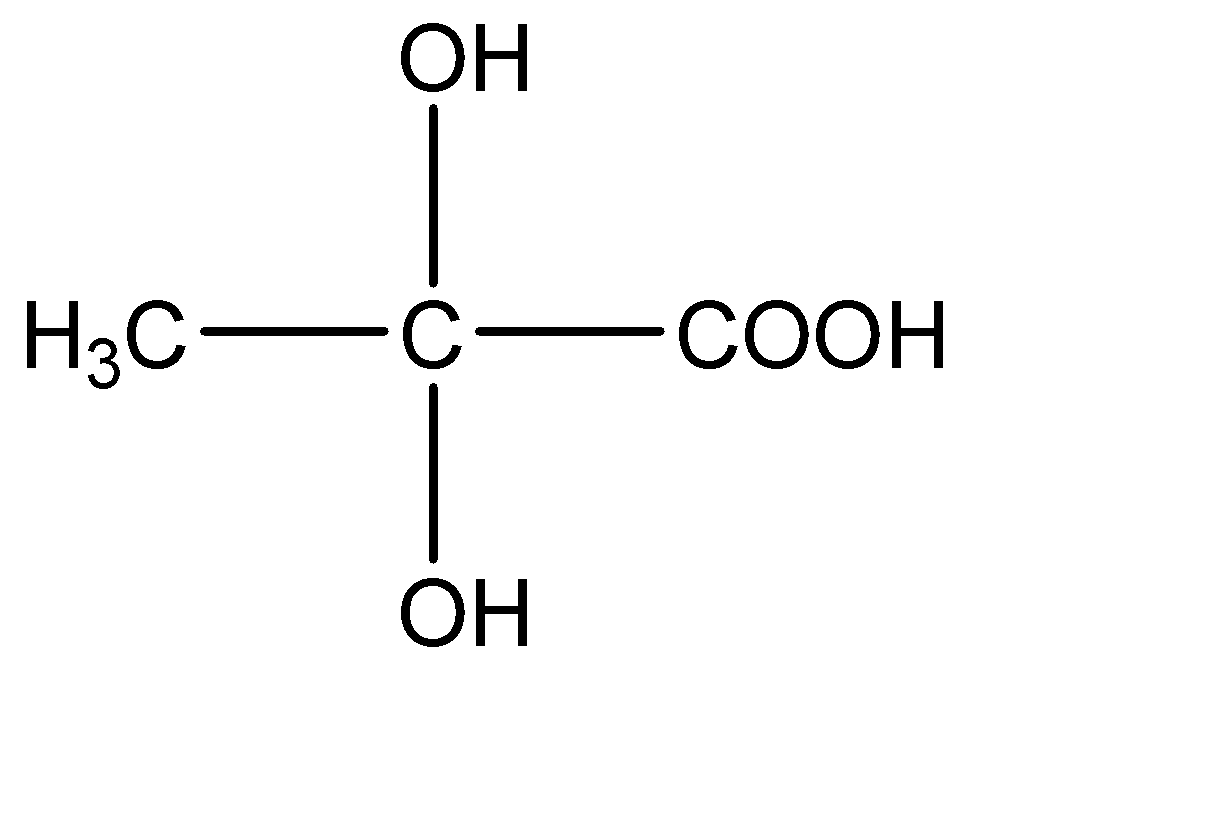
D.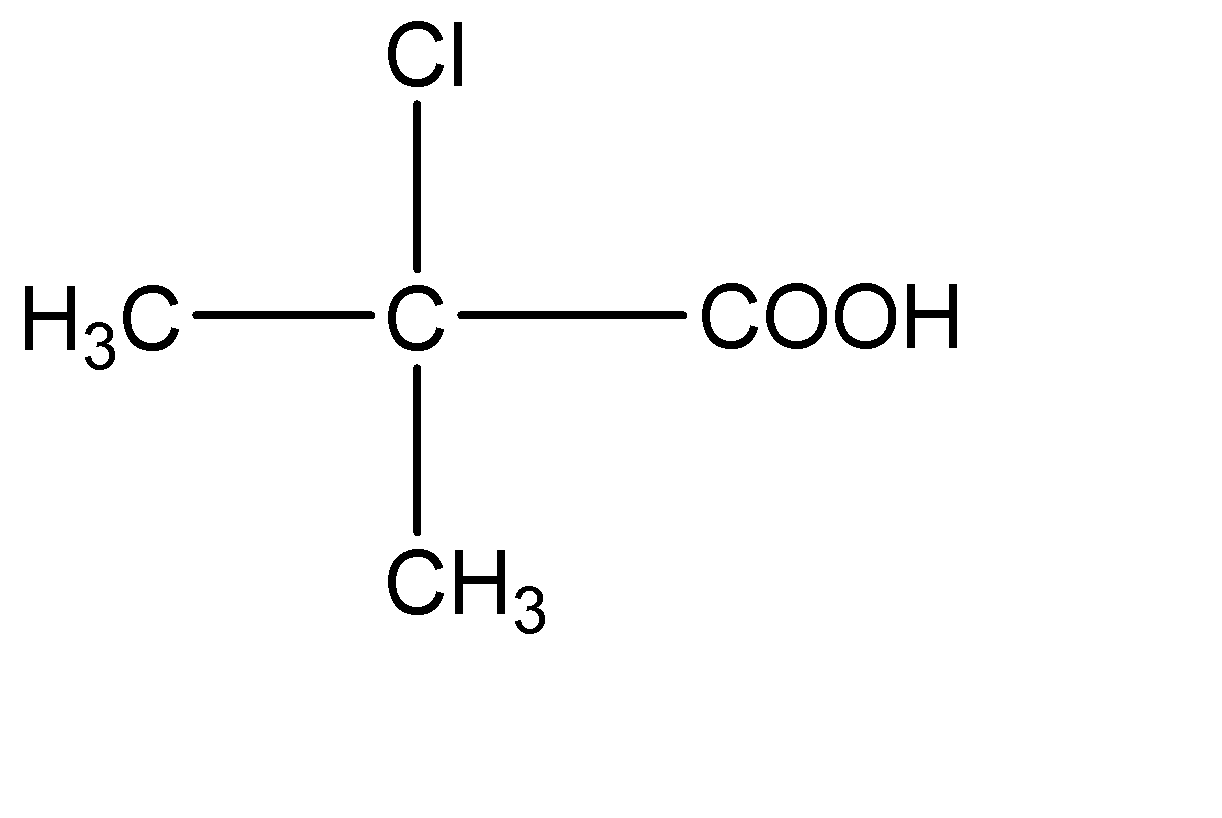
Answer
261.3k+ views
Hint: Optical activity can be understood as a phenomenon where the chiral carbon atoms in the organic compound tend to rotate the polarized light in either a clockwise or a counter – clockwise direction.
Complete Step-by-Step Answer:
Before we move forward with the solution of the given question, let us discuss some important basic concepts.
In an organic compound, a chiral carbon is the carbon atom which has four different atoms or molecules attached to it. It is important that all the four bonds formed by the carbon atom have different atoms or molecules for it to be called a chiral carbon. Chiral carbons are also responsible for a phenomenon known as optical activity. On the other hand, achiral carbons can be understood as the carbon atoms which have at least two bonded atoms or molecules to be the same. Achiral carbon atoms do not show optical activity.
Hence for finding the compound which shows optical activity from the given options, we must find the compound which has a chiral carbon.
1.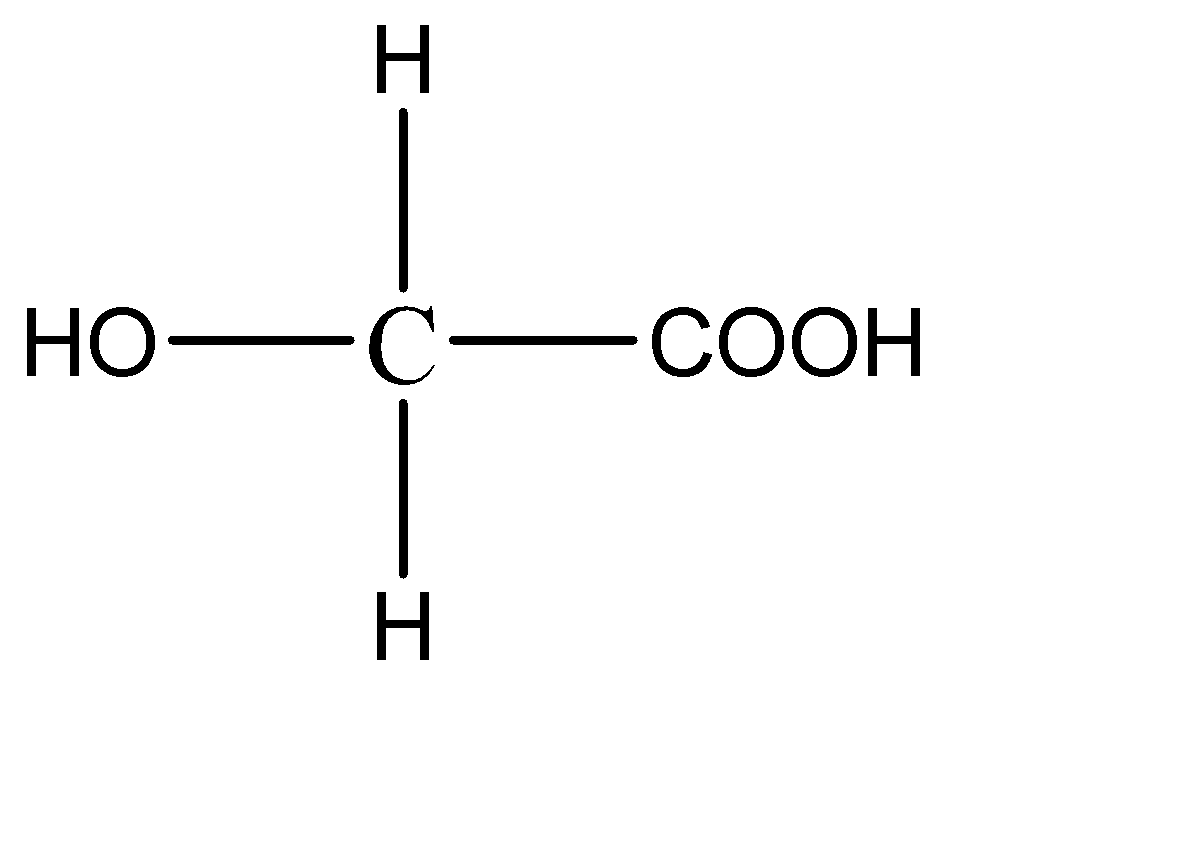
This compound has 2 hydrogen atoms on the central carbon atom. Hence this compound does not have a chiral carbon.
2.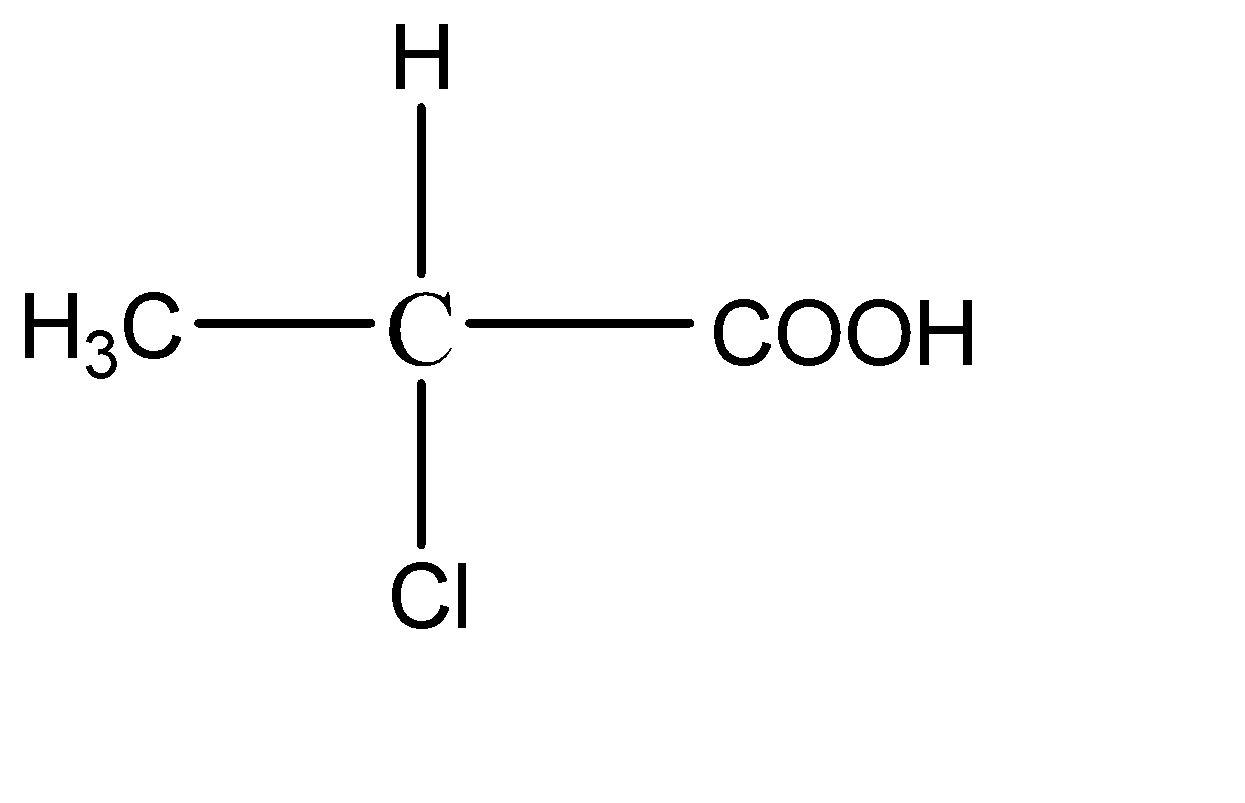
This compound has 4 different functional groups on all the bonds of the central carbon atom. Hence it has a chiral carbon.
3.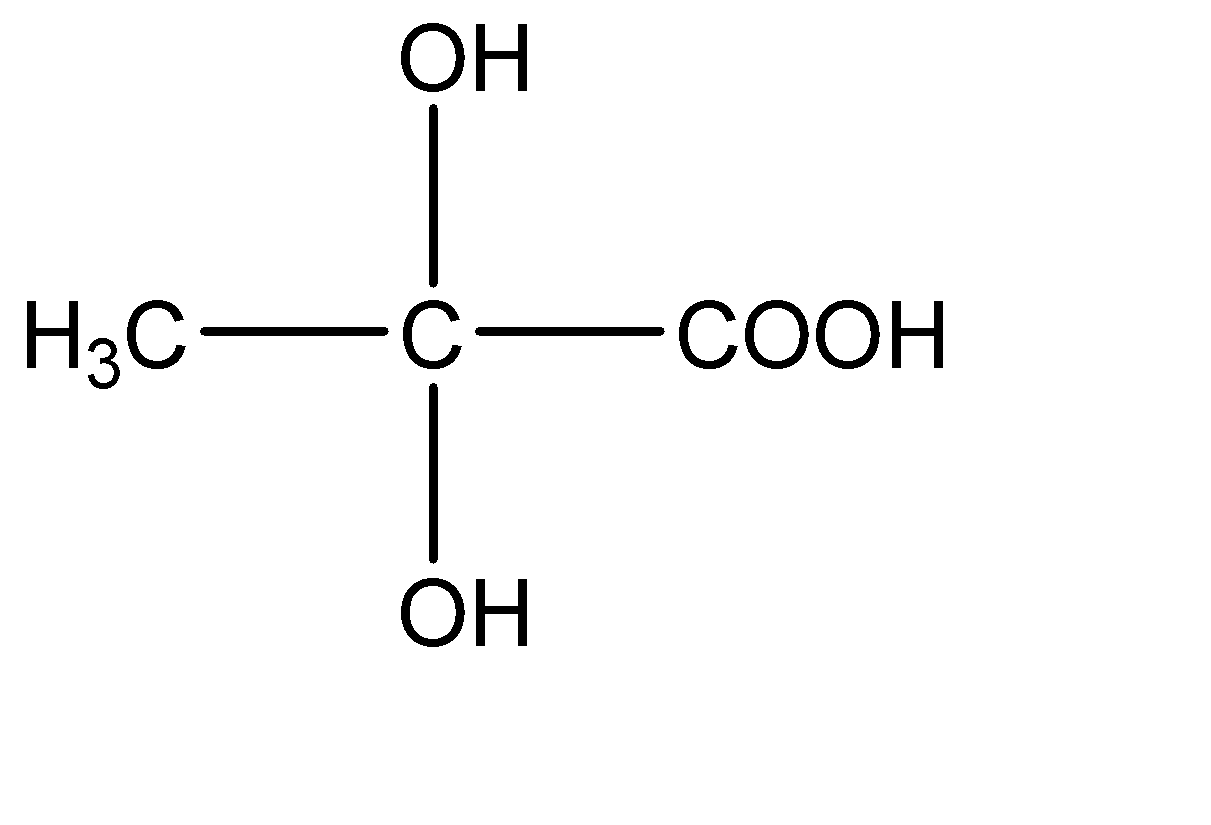
This compound has two -OH molecules on the central carbon atom. Hence this compound does not have a chiral carbon.
4.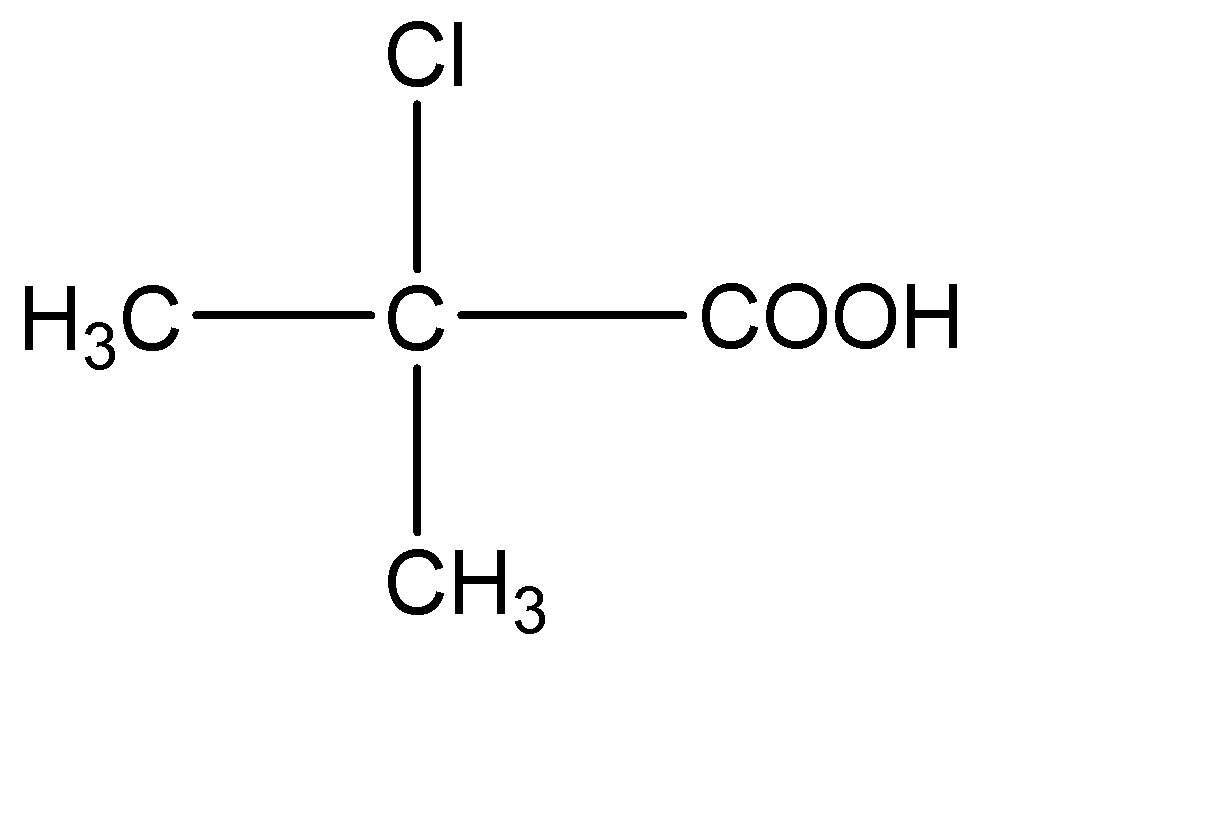
This compound has two - \[C{H_3}\] molecules on the central carbon atom. Hence this compound does not have a chiral carbon.
Hence, Option B is the correct option.
Note: In a solution if the total number of clockwise and counter – clockwise optical isomers is the same, then the solution is rendered optically inactive. This type of a mixture of optical isomers which is optically inactive is known as a racemic mixture.
Complete Step-by-Step Answer:
Before we move forward with the solution of the given question, let us discuss some important basic concepts.
In an organic compound, a chiral carbon is the carbon atom which has four different atoms or molecules attached to it. It is important that all the four bonds formed by the carbon atom have different atoms or molecules for it to be called a chiral carbon. Chiral carbons are also responsible for a phenomenon known as optical activity. On the other hand, achiral carbons can be understood as the carbon atoms which have at least two bonded atoms or molecules to be the same. Achiral carbon atoms do not show optical activity.
Hence for finding the compound which shows optical activity from the given options, we must find the compound which has a chiral carbon.
1.

This compound has 2 hydrogen atoms on the central carbon atom. Hence this compound does not have a chiral carbon.
2.

This compound has 4 different functional groups on all the bonds of the central carbon atom. Hence it has a chiral carbon.
3.

This compound has two -OH molecules on the central carbon atom. Hence this compound does not have a chiral carbon.
4.

This compound has two - \[C{H_3}\] molecules on the central carbon atom. Hence this compound does not have a chiral carbon.
Hence, Option B is the correct option.
Note: In a solution if the total number of clockwise and counter – clockwise optical isomers is the same, then the solution is rendered optically inactive. This type of a mixture of optical isomers which is optically inactive is known as a racemic mixture.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




