Which of the following is allylic halide?
(a) (1-Bromoethyl) benzene
(b) Benzyl chloride
(c) 1-Bromo benzene
(d) 3-Chloro cyclohex-1-ene
Answer
264.6k+ views
Hint: An allylic carbon is a carbon atom bonded to a carbon atom that in turn is doubly bonded to another carbon atom.
Step-by-step answer:
> In (1-Bromoethyl) benzene or \[{{C}_{8}}{{H}_{9}}Br\], we can see that on the allylic carbon of the molecule, the halogen atom is present with a methyl group. Hence, it cannot be termed as an allylic halide.
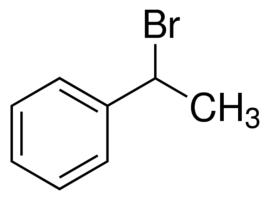
(1-Bromoethyl) benzene
> In Benzyl chloride or (Chloromethyl) Benzene with a molecular formula of \[{{C}_{7}}{{H}_{7}}Cl\], we can see that on the allylic carbon of the molecule, the halogen atom is present along with a methyl group. This is why Benzyl chloride does not qualify as an allylic halide.
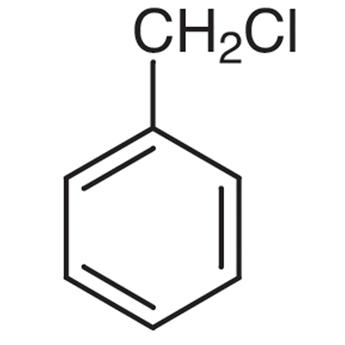
Benzyl chloride
> 1-Bromo benzene or \[{{C}_{6}}{{H}_{5}}Br\] as we can see does not have the halogen atom Br bonded to \[s{{p}^{3}}-hybridised\] carbon atom next to the Carbon-Carbon double bond \[\left( C=C \right)\]. This denotes that 1-Bromo benzene is not an allylic halide.
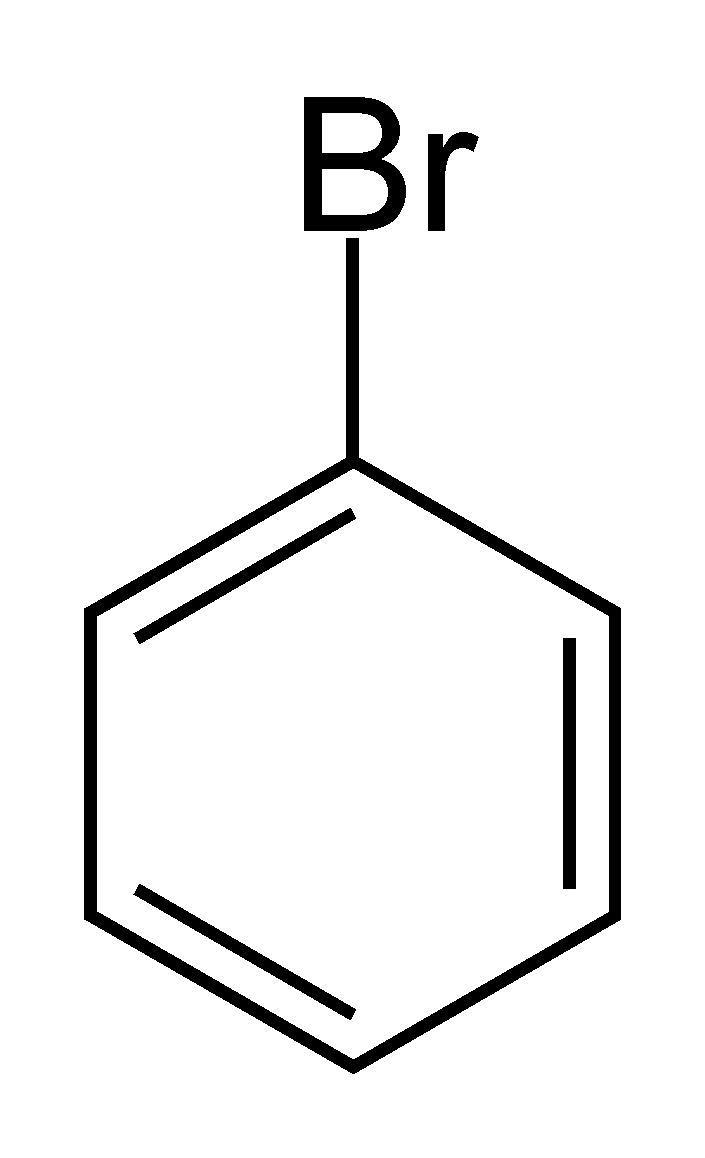
1-Bromo benzene
> An allylic halide is an alkyl halide which has one or more halogen atoms on the allylic carbons of the molecule. In the below given figure of 3-Chloro cyclohex-1-ene, we can see a halogen atom bonded to \[s{{p}^{3}}-hybridised\] carbon atom next to the Carbon- Carbon double bond \[\left( C=C \right)\]. This denotes that 3-Chloro cyclohex-1-ene or \[{{C}_{7}}{{H}_{11}}Cl\] is allylic halide.
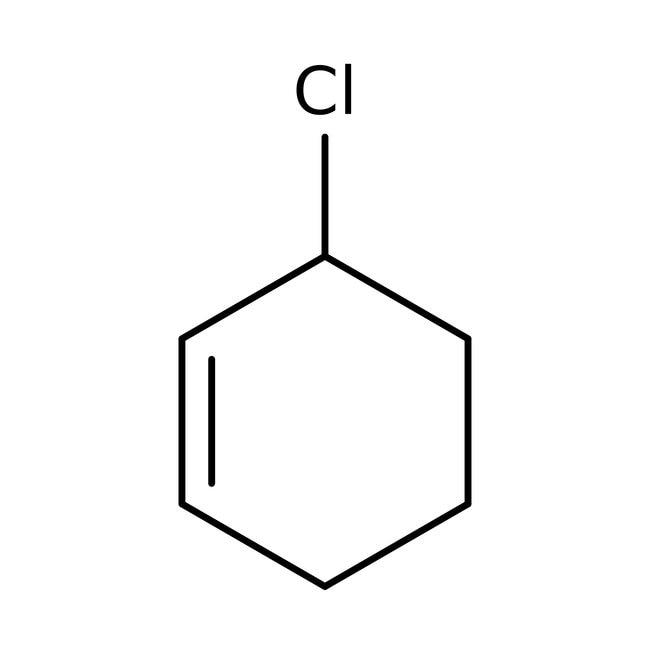
3-Chloro cyclohex-1-ene
So, we can figure out that Option D is the correct answer.
Note: Allylic halide is the one in which the carbon atom next to the double bonded carbon atom carries one or more halogen atoms. Allylic halides are reactive in both \[{{S}_{N}}1\] and \[{{S}_{N}}2\] mechanisms.
Step-by-step answer:
> In (1-Bromoethyl) benzene or \[{{C}_{8}}{{H}_{9}}Br\], we can see that on the allylic carbon of the molecule, the halogen atom is present with a methyl group. Hence, it cannot be termed as an allylic halide.

(1-Bromoethyl) benzene
> In Benzyl chloride or (Chloromethyl) Benzene with a molecular formula of \[{{C}_{7}}{{H}_{7}}Cl\], we can see that on the allylic carbon of the molecule, the halogen atom is present along with a methyl group. This is why Benzyl chloride does not qualify as an allylic halide.

Benzyl chloride
> 1-Bromo benzene or \[{{C}_{6}}{{H}_{5}}Br\] as we can see does not have the halogen atom Br bonded to \[s{{p}^{3}}-hybridised\] carbon atom next to the Carbon-Carbon double bond \[\left( C=C \right)\]. This denotes that 1-Bromo benzene is not an allylic halide.

1-Bromo benzene
> An allylic halide is an alkyl halide which has one or more halogen atoms on the allylic carbons of the molecule. In the below given figure of 3-Chloro cyclohex-1-ene, we can see a halogen atom bonded to \[s{{p}^{3}}-hybridised\] carbon atom next to the Carbon- Carbon double bond \[\left( C=C \right)\]. This denotes that 3-Chloro cyclohex-1-ene or \[{{C}_{7}}{{H}_{11}}Cl\] is allylic halide.

3-Chloro cyclohex-1-ene
So, we can figure out that Option D is the correct answer.
Note: Allylic halide is the one in which the carbon atom next to the double bonded carbon atom carries one or more halogen atoms. Allylic halides are reactive in both \[{{S}_{N}}1\] and \[{{S}_{N}}2\] mechanisms.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




