Which of the following is a non-reducing sugar?
A. Glucose
B. Sucrose
C. Maltose
D. Lactose
Answer
266.1k+ views
Hint: Non-reducing sugars does have a group attached to any of the anomeric carbon. Therefore, they are unable to reduce other compounds.
Complete step by step answer:
We can draw the structure of sucrose as follows:
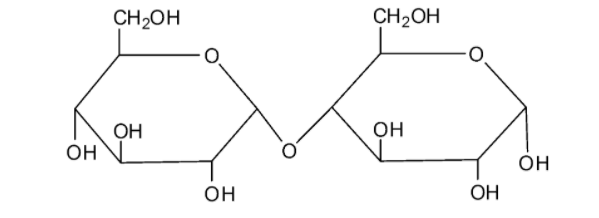
The molecule of sucrose is a disaccharide. From the structure, we can see that sucrose is a combination of the monosaccharides fructose and glucose with the formula ${{\rm{C}}_{{\rm{12}}}}{{\rm{H}}_{{\rm{22}}}}{{\rm{O}}_{{\rm{11}}}}$. It is a non-reducing sugar as this molecule does not have characteristics of the reducing sugars. The two monosaccharide units are connected by a glycosidic linkage between ${{\rm{C}}_{\rm{1}}}$ of ${\rm{\alpha }} - $ glucose and ${{\rm{C}}_{\rm{2}}}$ of ${\rm{\beta }} - $ fructose. As the reducing groups of the glucose molecule and fructose molecules are involved in the formation of the glycosidic, sucrose is considered a non-reducing sugar.
Therefore, Sucrose is a non-reducing sugar.
So, out of the given four options, B is the correct option.
Additional information:
We can draw the structure of glucose as follows:
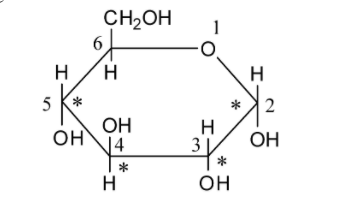
Glucose is a monosaccharide. As glucose acts as a reducing agent, it is reconsidered as a reducing sugar.
We can draw the structure of maltose as follows:
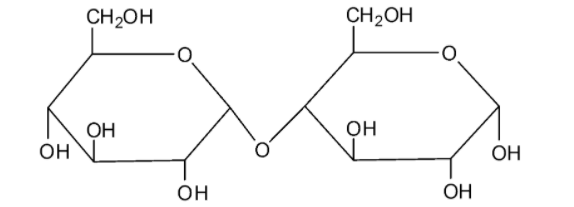
Maltose is made by the combination of two glucose molecules. Maltose undergoes mutarotation. Because of this reason, maltose is considered as a reducing sugar.
We can draw the structure of lactose as follows:
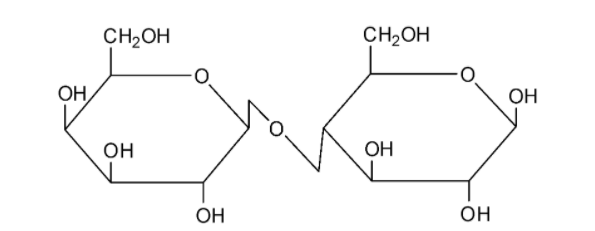
Lactose is composed by the combination of a glucose and a galactose molecule. Lactose undergoes mutarotation and it is hence a reducing sugar.
Note:
Monosaccharides are considered as reducing sugars. Then sugar that cannot act as a reducing agent are non-reducing agents. Out of disaccharides, sucrose is a well-known non-reducing sugar.
Complete step by step answer:
We can draw the structure of sucrose as follows:

The molecule of sucrose is a disaccharide. From the structure, we can see that sucrose is a combination of the monosaccharides fructose and glucose with the formula ${{\rm{C}}_{{\rm{12}}}}{{\rm{H}}_{{\rm{22}}}}{{\rm{O}}_{{\rm{11}}}}$. It is a non-reducing sugar as this molecule does not have characteristics of the reducing sugars. The two monosaccharide units are connected by a glycosidic linkage between ${{\rm{C}}_{\rm{1}}}$ of ${\rm{\alpha }} - $ glucose and ${{\rm{C}}_{\rm{2}}}$ of ${\rm{\beta }} - $ fructose. As the reducing groups of the glucose molecule and fructose molecules are involved in the formation of the glycosidic, sucrose is considered a non-reducing sugar.
Therefore, Sucrose is a non-reducing sugar.
So, out of the given four options, B is the correct option.
Additional information:
We can draw the structure of glucose as follows:

Glucose is a monosaccharide. As glucose acts as a reducing agent, it is reconsidered as a reducing sugar.
We can draw the structure of maltose as follows:

Maltose is made by the combination of two glucose molecules. Maltose undergoes mutarotation. Because of this reason, maltose is considered as a reducing sugar.
We can draw the structure of lactose as follows:

Lactose is composed by the combination of a glucose and a galactose molecule. Lactose undergoes mutarotation and it is hence a reducing sugar.
Note:
Monosaccharides are considered as reducing sugars. Then sugar that cannot act as a reducing agent are non-reducing agents. Out of disaccharides, sucrose is a well-known non-reducing sugar.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




