Which of the following is a monobasic acid?
A) \[{H_2}S{O_4}\]
B) \[C{H_3}COOH\]
C) \[{H_3}P{O_3}\]
D) \[{H_2}C{O_3}\]
Answer
261.6k+ views
Hint: The basicity of acid is defined as the number of replaceable hydrogen present in an acid molecule. So, by the ionization of all the given acid molecules, the one which does not replace more than one hydrogen will be the monobasic acid.
Complete step by step answer:
Let us first see the structure of \[{H_2}S{O_4}\].
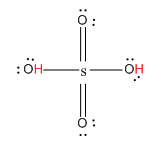
Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are two transferable hydrogen atoms, \[{H_2}S{O_4}\] is the dibasic acid.
Now, let us see the structure of \[{H_3}P{O_3}\].
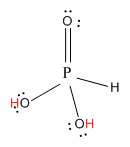
Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are two transferable hydrogen atoms, \[{H_3}P{O_3}\] is the dibasic acid.
Now, let us see the structure of \[{H_2}C{O_3}\].
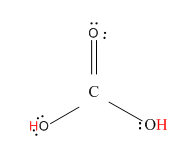
Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are two transferable hydrogen atoms, \[{H_2}C{O_3}\] is the dibasic acid.
Now, let us see the structure of \[C{H_3}COOH\].
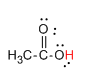
Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are only one transferable hydrogen atom, \[C{H_3}COOH\] is the monobasic acid.
We can also show the ionizable hydrogen atom by dissociation of the acetic acid molecule as follows.
\[C{H_3}COOH \to C{H_3}CO{O^ - } + {H^ + }\]
The above reaction shows only one ionizable hydrogen atom. So, its basicity is also one.
Therefore, we can conclude that the correct answer to this question is option B.
Note:
The hydrogen attached to the oxygen is only replaced and the hydrogen attached to the central atom is irreplaceable as in phosphorous acid and acetic acid. Always focus on the structure for this type of question.
Complete step by step answer:
Let us first see the structure of \[{H_2}S{O_4}\].

Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are two transferable hydrogen atoms, \[{H_2}S{O_4}\] is the dibasic acid.
Now, let us see the structure of \[{H_3}P{O_3}\].

Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are two transferable hydrogen atoms, \[{H_3}P{O_3}\] is the dibasic acid.
Now, let us see the structure of \[{H_2}C{O_3}\].

Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are two transferable hydrogen atoms, \[{H_2}C{O_3}\] is the dibasic acid.
Now, let us see the structure of \[C{H_3}COOH\].

Here, the hydrogen marked with red color is the transferrable hydrogen atom. As there are only one transferable hydrogen atom, \[C{H_3}COOH\] is the monobasic acid.
We can also show the ionizable hydrogen atom by dissociation of the acetic acid molecule as follows.
\[C{H_3}COOH \to C{H_3}CO{O^ - } + {H^ + }\]
The above reaction shows only one ionizable hydrogen atom. So, its basicity is also one.
Therefore, we can conclude that the correct answer to this question is option B.
Note:
The hydrogen attached to the oxygen is only replaced and the hydrogen attached to the central atom is irreplaceable as in phosphorous acid and acetic acid. Always focus on the structure for this type of question.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

Understanding Atomic Structure for Beginners




