Which of the following compounds is known as inorganic benzene?
A. ${{B}_{2}}{{H}_{6}}$
B. ${{C}_{5}}{{H}_{5}}B$
C. ${{C}_{3}}{{N}_{3}}{{H}_{3}}$
D. ${{B}_{3}}{{N}_{3}}{{H}_{6}}$
Answer
261.6k+ views
Hint: Inorganic benzene is a cyclic compound, it is called as inorganic benzene because it is isoelectronic and also isostructural with benzene. It is actually a colourless liquid.
Step by step solution:
Borazine is called as the inorganic benzene, which is having chemical formula ${{B}_{3}}{{N}_{3}}{{H}_{6}}$. It is a cyclic compound , we can see the structure of borazine:
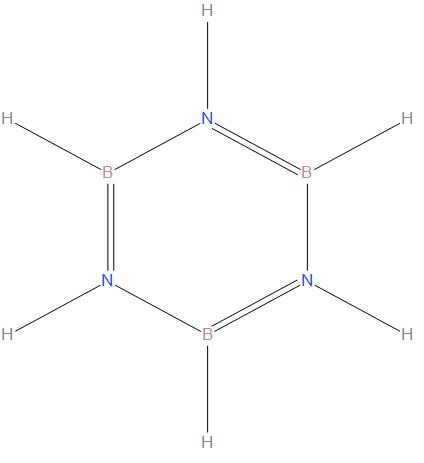
- The bond angle is found to be ${{117.1}^{\circ }}$ at the boron atoms and ${{122.9}^{\circ }}$ at the nitrogen, which will give the distinct symmetry to the molecule.
- There are several properties of borazine found like:
- It is isoelectronic and also isostructural with benzene, isoelectronic means those which have the same number of electrons around them and isostructural means those that are having the same hybridisation and shape as that of others.
-It is colourless and also has an aromatic smell.
- In borazine 12 sigma bonds and 3 pi bonds are found that are the same as in benzene.
- Now let’s see how borazine is being prepared: One of the main method of preparation of borazine is from diborane and ammonia, which are taken in a ratio of 1:2 at about temperature of ${{300}^{\circ }}C$. The reaction can be written as:
\[3{{B}_{2}}{{H}_{6}}+6N{{H}_{3}}\to 2{{B}_{3}}{{H}_{6}}{{N}_{3}}+12{{H}_{2}}\]
-The are several uses of borazine found like:
-It can be used as a precursor for growing boron nitride thin films on various surfaces.
- It is found that these are also used as a starting material for the various other ceramics like boron carbonitrides.
Hence, we can conclude that the correct option is (D) ,${{B}_{3}}{{N}_{3}}{{H}_{6}}$ is known as inorganic benzene.
Note:
We must remember that borazine is also called as borazole along with inorganic benzene, which is a polar compound, in which there are three BH units and three NH units are present alternate.
Step by step solution:
Borazine is called as the inorganic benzene, which is having chemical formula ${{B}_{3}}{{N}_{3}}{{H}_{6}}$. It is a cyclic compound , we can see the structure of borazine:

- The bond angle is found to be ${{117.1}^{\circ }}$ at the boron atoms and ${{122.9}^{\circ }}$ at the nitrogen, which will give the distinct symmetry to the molecule.
- There are several properties of borazine found like:
- It is isoelectronic and also isostructural with benzene, isoelectronic means those which have the same number of electrons around them and isostructural means those that are having the same hybridisation and shape as that of others.
-It is colourless and also has an aromatic smell.
- In borazine 12 sigma bonds and 3 pi bonds are found that are the same as in benzene.
- Now let’s see how borazine is being prepared: One of the main method of preparation of borazine is from diborane and ammonia, which are taken in a ratio of 1:2 at about temperature of ${{300}^{\circ }}C$. The reaction can be written as:
\[3{{B}_{2}}{{H}_{6}}+6N{{H}_{3}}\to 2{{B}_{3}}{{H}_{6}}{{N}_{3}}+12{{H}_{2}}\]
-The are several uses of borazine found like:
-It can be used as a precursor for growing boron nitride thin films on various surfaces.
- It is found that these are also used as a starting material for the various other ceramics like boron carbonitrides.
Hence, we can conclude that the correct option is (D) ,${{B}_{3}}{{N}_{3}}{{H}_{6}}$ is known as inorganic benzene.
Note:
We must remember that borazine is also called as borazole along with inorganic benzene, which is a polar compound, in which there are three BH units and three NH units are present alternate.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

Understanding Atomic Structure for Beginners




