Which of the following compounds can give a cannizzaro reaction?
A.
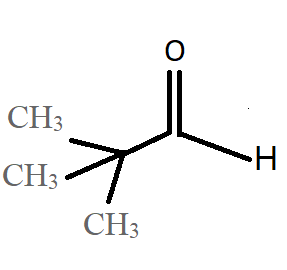
B.
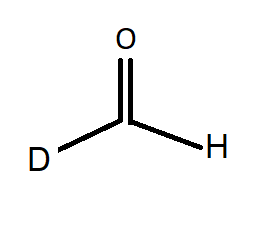
C.
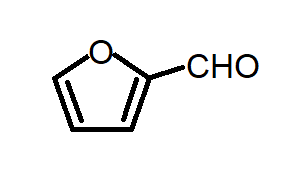
D. All of the above
Answer
249.9k+ views
Hint: Cannizaro reaction is a disproportionation reaction which involves aldehyde with no alpha hydrogen as the reactant. The aldehyde group oxidises and reduces itself to give primary alcohol and a carboxylic acid as major products. Cannizaro reaction takes place in presence of a strong base like $NaOH$followed by \[{H^ + }\]. Cross Cannizaro reaction is not an example of disproportionation but a redox reaction
Complete Step by Step Solution:
Option A: trimethylacetaldehyde or also known as pivalaldehyde has formula \[{C_5}{H_{10}}O\]. Aldehyde group has hydrogen on one side and trimethyl on the other hence, there is no alpha hydrogen. Thus, the product can undergo Cannizaro reaction to form trimethyl acetic acid and 1.1-dimethyl ethanol.
Option B: formaldehyde has chemical formula $HCHO$. Formaldehyde has no alpha hydrogen hence undergoes Cannizaro reaction. If one of the hydrogen is replaced by deuterium then it has no effect on the reaction. Hence the given compound also gives Cannizaro reaction.
Option C: the given compound is known as furfural which is an aldehyde group substituted at the $2$ position of furan. Furfural undergoes Cannizaro reaction and gives furoic acid and furfuryl alcohol as major products.
Option D: thus, all the given compounds give Cannizaro because of absence of any alpha position. So, the correct answer is D.
Note: In the figure below there are two positions from aldehyde group $C = 0$ marked as a and b. The position adjacent to the aldehyde group marked as ‘a’ is known as the alpha position. The next position marked as ‘b’ is known as beta position. Thus, the number of alpha hydrogen in the compound is $5$ and $3$ beta hydrogen.
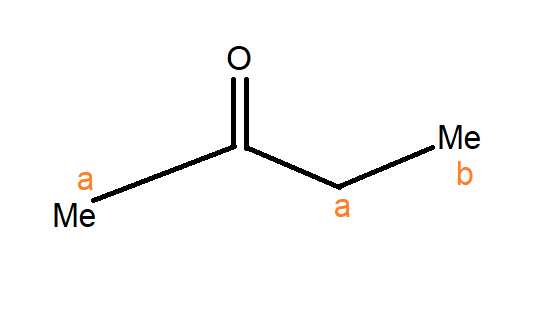
Complete Step by Step Solution:
Option A: trimethylacetaldehyde or also known as pivalaldehyde has formula \[{C_5}{H_{10}}O\]. Aldehyde group has hydrogen on one side and trimethyl on the other hence, there is no alpha hydrogen. Thus, the product can undergo Cannizaro reaction to form trimethyl acetic acid and 1.1-dimethyl ethanol.
Option B: formaldehyde has chemical formula $HCHO$. Formaldehyde has no alpha hydrogen hence undergoes Cannizaro reaction. If one of the hydrogen is replaced by deuterium then it has no effect on the reaction. Hence the given compound also gives Cannizaro reaction.
Option C: the given compound is known as furfural which is an aldehyde group substituted at the $2$ position of furan. Furfural undergoes Cannizaro reaction and gives furoic acid and furfuryl alcohol as major products.
Option D: thus, all the given compounds give Cannizaro because of absence of any alpha position. So, the correct answer is D.
Note: In the figure below there are two positions from aldehyde group $C = 0$ marked as a and b. The position adjacent to the aldehyde group marked as ‘a’ is known as the alpha position. The next position marked as ‘b’ is known as beta position. Thus, the number of alpha hydrogen in the compound is $5$ and $3$ beta hydrogen.

Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




