Two charges of 10 C and -15 C are separated in air by 1m. The ratio of magnitude of force exerted by one on the other is:
A) 1:2
B) 2:1
C) 1:1
D) None of these
Answer
253.5k+ views
Hint: Coulomb's force between the two point charges follows Newton's third law of which states that, two mutually interacting particles have oppositely directed forces on each other.
Complete step by step solution:
Electrostatic force is an action reaction pair and hence two charged particles exert equal and opposite forces on each other despite differences in the magnitude of the individual charges. (The strength of this force is governed by Coulomb's law.)
Therefore, The ratio of magnitudes of mutual coulomb force acting on the two point charges is simply 1 : 1.
Option C is correct.
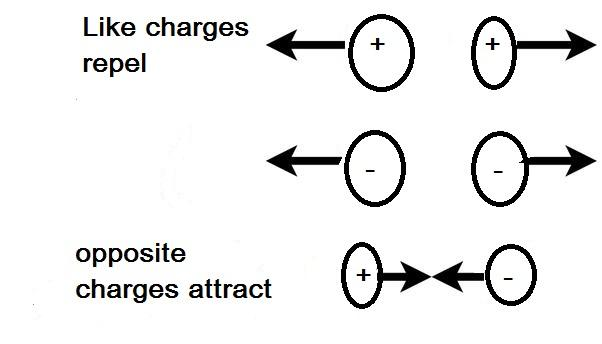
Additional Information: Electric charge is the property of objects that gives rise to this observed force. Like gravity, electric force "acts at a distance". The electric force is much larger than the gravity force. Unlike gravity, there are two types of electric charge , Unlike charges attract, Like charges repel each other. There is only one type of gravity, gravity only attracts.
The force exerted by one charge q on another charge Q is given by Coulomb's law:
According to Coulomb's law, the force of attraction or repulsion between two charged bodies is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. This point works along the line joining the two charges considered to be charges.
Note: Remember that force is a vector quantity, so when more than one charge exerts a force on another charge, the net force on that charge is the vector sum of the individual forces. Remember, too, that charges of the same signal exert repulsive forces on one another, while charges of the opposite sign attract.
Complete step by step solution:
Electrostatic force is an action reaction pair and hence two charged particles exert equal and opposite forces on each other despite differences in the magnitude of the individual charges. (The strength of this force is governed by Coulomb's law.)
Therefore, The ratio of magnitudes of mutual coulomb force acting on the two point charges is simply 1 : 1.
Option C is correct.
Additional Information: Electric charge is the property of objects that gives rise to this observed force. Like gravity, electric force "acts at a distance". The electric force is much larger than the gravity force. Unlike gravity, there are two types of electric charge , Unlike charges attract, Like charges repel each other. There is only one type of gravity, gravity only attracts.

The force exerted by one charge q on another charge Q is given by Coulomb's law:
According to Coulomb's law, the force of attraction or repulsion between two charged bodies is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. This point works along the line joining the two charges considered to be charges.
Note: Remember that force is a vector quantity, so when more than one charge exerts a force on another charge, the net force on that charge is the vector sum of the individual forces. Remember, too, that charges of the same signal exert repulsive forces on one another, while charges of the opposite sign attract.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Young’s Double Slit Experiment Derivation Explained

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Wheatstone Bridge – Principle, Formula, Diagram & Applications

Circuit Switching vs Packet Switching: Key Differences Explained

Mass vs Weight: Key Differences Explained for Students

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
CBSE Class 12 Physics Question Paper 2026: Download SET-wise PDF with Answer Key & Analysis

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score




