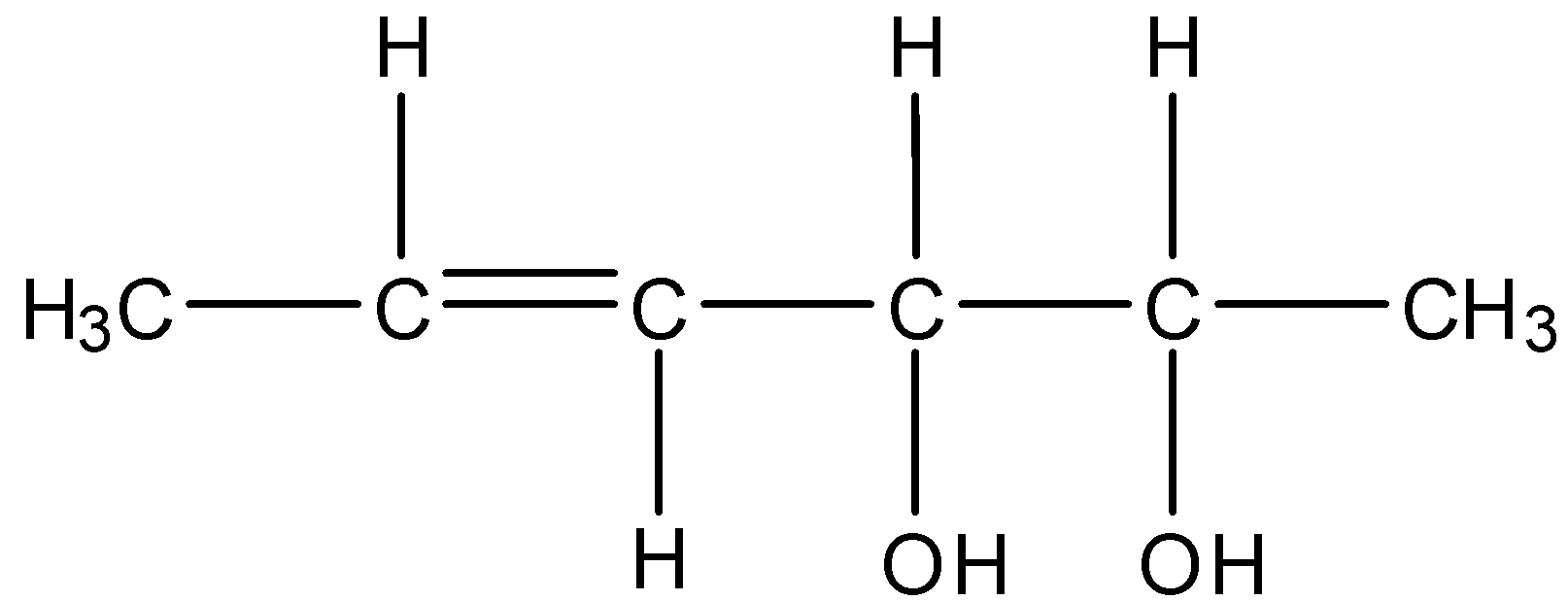
The total number of configurational isomers of the given compound are:
A. 2
B. 4
C. 6
D. 8
Answer
256.8k+ views
Hint: Configurational isomers are the stereoisomers that cannot be converted into one another by rotation around a single bond in the molecule. There are two types of Configurational isomers. 1) Geometrical isomers and 2) optical isomers.
Complete step by step solution:
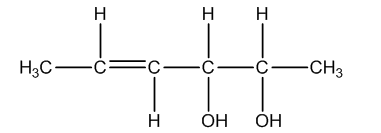
Total number of configurational isomers = geometrical isomers + optical isomers.
So, totally 8 configurational isomers are going to form by the given compound.
So, the correct option is D.
Note: Don’t be confused between geometrical isomerism and optical isomerism. The compounds that have double bonds will show geometrical isomerism and the compounds which have chiral centers show the optical isomerism. Both the isomerisms combined are called geometrical isomerism.
Complete step by step solution:
Geometrical isomerism is going to show by unsaturated compounds and optical isomerism is going to exhibit by the molecules that are having chiral carbons.
The given molecule contains six carbons, one double bond and two chiral centers.

Now we will talk about geometrical isomerism.
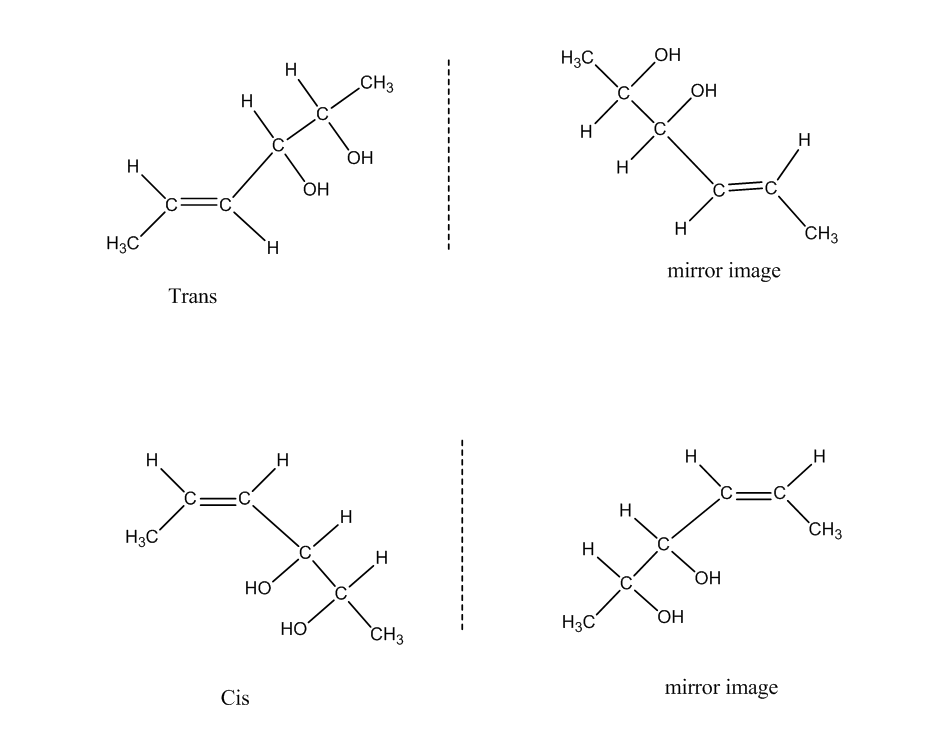
The following are the geometrical isomers of the given compound.

Now there are two forms called Trans and Cis they have mirror images also. So, the geometrical isomers formed are four.
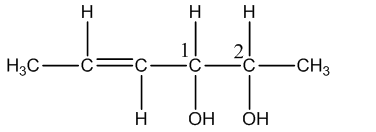
Coming to optical isomers, the given compound has two chiral centers and shown in the below image.

The numbers 1 and 2 represent two chiral centers.
There is a formula to calculate the number of isomers from chiral centers.
Optical isomers = \[{{2}^{n}}\], here n = number of chiral centers.
We know that in the given compound there are two chiral centers.
Therefore the number of optical isomers = \[{{2}^{2}}=4\].
Total number of configurational isomers = geometrical isomers + optical isomers.
= 4 +4
= 8.
So, totally 8 configurational isomers are going to form by the given compound.
So, the correct option is D.
Note: Don’t be confused between geometrical isomerism and optical isomerism. The compounds that have double bonds will show geometrical isomerism and the compounds which have chiral centers show the optical isomerism. Both the isomerisms combined are called geometrical isomerism.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main Mock Test 2025-26: Electromagnetic Induction & Alternating Currents

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Cut-Off for MNIT Jaipur 2025: Analysis and Important Details

JEE Main 2025 Cut-off For NIT Andhra Pradesh

Other Pages
CBSE Class 12 Physics Question Paper 2026: Download SET-wise PDF with Answer Key & Analysis

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Accountancy Question Paper 2026 (24 February) – Answer Key and PDF Download

CBSE Class 12 Political Science Important Questions (2025-26)

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26




