The monomer used in Novolac; a polymer used in paints is:
A. Butadiene and styrene
B. Phenol and formaldehyde
C. Butadiene and acrylo nitrile
D. Melamine and formaldehyde
Answer
268.8k+ views
Hint: There are different classes of polymers. Novolac is a phenolic resin type of polymer. It is extensively used in microelectronics.
Complete step by step answer: A polymer is a large molecule formed by continuous linking of many small subunits called Monomers. Basically, it is a repetition of monomers.
Another name for novolac is phenolic resin or phenol formaldehyde resin. As this suggests, the two monomers needed to form novolac are phenol and formaldehyde.
The reaction that occurs is –
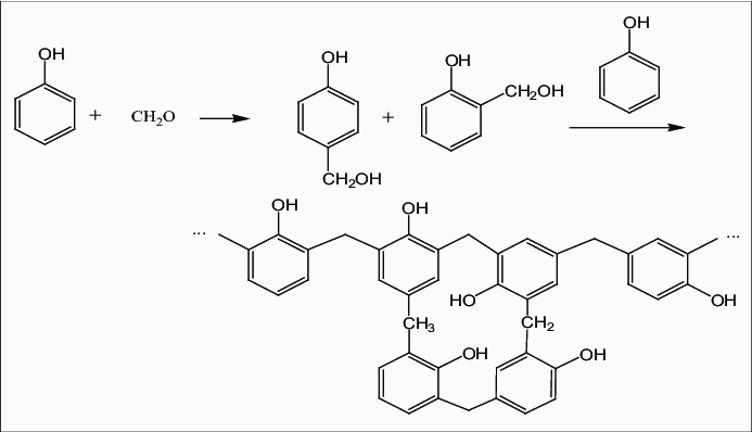
The reaction stops when the formaldehyde reactant is exhausted, often leaving up to 10% of un-reacted phenol.
So, B is the correct option.
Additional information: There are two types of polymers, natural and artificial. Natural polymers are found in plants. Apart from natural and manmade, polymers are classified in various other ways.
Based on the type of monomer used and mechanism of polymerization, there are different types of polymers. Novolac is a cross linked polymer formed by condensation polymerisation. During preparation of novolac, as a general rule phenol gets substituted on both ortho and para positions. But we know that para position is almost twice as reactive as ortho. But there are twice as many ortho sites (two per phenol molecule) so the fractions of ortho-ortho, para-para and ortho-para bridges are approximately equal. In this reaction, a water molecule gets eliminated. Hence the type of polymerization mechanism is condensation. Distillation of the molten resin during manufacturing removes the excess phenol and water.
Note: To remember this easily, we can keep in mind that novolac is a phenolic resin type of polymer. It has a cross linked structure and is formed by condensation polymerization.
Complete step by step answer: A polymer is a large molecule formed by continuous linking of many small subunits called Monomers. Basically, it is a repetition of monomers.
Another name for novolac is phenolic resin or phenol formaldehyde resin. As this suggests, the two monomers needed to form novolac are phenol and formaldehyde.
The reaction that occurs is –

The reaction stops when the formaldehyde reactant is exhausted, often leaving up to 10% of un-reacted phenol.
So, B is the correct option.
Additional information: There are two types of polymers, natural and artificial. Natural polymers are found in plants. Apart from natural and manmade, polymers are classified in various other ways.
Based on the type of monomer used and mechanism of polymerization, there are different types of polymers. Novolac is a cross linked polymer formed by condensation polymerisation. During preparation of novolac, as a general rule phenol gets substituted on both ortho and para positions. But we know that para position is almost twice as reactive as ortho. But there are twice as many ortho sites (two per phenol molecule) so the fractions of ortho-ortho, para-para and ortho-para bridges are approximately equal. In this reaction, a water molecule gets eliminated. Hence the type of polymerization mechanism is condensation. Distillation of the molten resin during manufacturing removes the excess phenol and water.
Note: To remember this easily, we can keep in mind that novolac is a phenolic resin type of polymer. It has a cross linked structure and is formed by condensation polymerization.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

Dynamic Equilibrium – Definition, Examples, Henry’s Law and FAQs

JEE General Topics in Chemistry Important Concepts and Tips

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Kinematics Mock Test for JEE Main 2025-26: Comprehensive Practice

Vector Algebra Mock Test 1-2 for JEE Main 2025-26 Preparation

JEE Main Cut-Off for MNIT Jaipur 2025: Analysis and Important Details

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
CBSE Class 12 Physics Question Paper 2026: Download SET-wise PDF with Answer Key & Analysis

JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Accountancy Question Paper 2026 (24 February) – Answer Key and PDF Download

CBSE Class 12 Political Science Important Questions (2025-26)

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key




