The molecular shapes of \[S{{F}_{4}}\], \[C{{F}_{4}}\]and \[Xe{{F}_{4}}\] are:
A.The same, with 1, 0 and 2 lone pair of electrons respectively
B.The same, with 1, 1 and 1 lone pair of electrons respectively
C.Different, with 0, 1 and 2 lone pair of electrons respectively
D.Different, with 1,0 and 2 lone pair of electrons respectively
Answer
260.1k+ views
Hint: For finding out the lone pair of electrons and the molecular shapes of the compounds, you need to find out the electrons in the valence shell. The total valence electron contribution is to be counted from the valence electrons of the atoms of the compound and the difference of it from the nearest noble gas configuration gives the number of lone pairs of electrons.
Complete step by step answer:
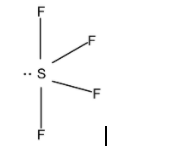
\[S{{F}_{4}}\]:-
Valency of sulphur = 6
Valency of fluorine = 7
So, the total valence electron contribution = \[[6+(7\times 4)]\]= 34 electrons
The nearest multiple of noble gas is 32.
So, (34-32) = 2 electrons exist as a lone pair on the sulphur atom.
Now, the sulphur forms four single bonds to the fluorine atoms. Each fluorine atom has three lone pairs to complete their octet.
Since a lone pair experiences repulsion, it is placed in equatorial position to reduce repulsion. Sulphur forms four single bonds and one lone pair. So, the hybridisation is \[\text{s}{{\text{p}}^{3}}\text{d}\]. The molecular shape is trigonal bipyramidal.
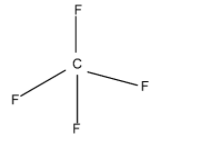
\[C{{F}_{4}}\]:-
Valency of carbon = 4
Valency of fluorine = 7
So, the total valence electron contribution = \[[4+(7\times 4)]\]= 32 electrons
The nearest multiple of noble gas is 32.
So, there is no lone pair of electrons on the carbon atom. The hybridisation is \[\text{s}{{\text{p}}^{3}}\]. The molecular shape is tetrahedral.
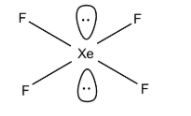
\[Xe{{F}_{4}}\]:-
Valency of xenon = 8
Valency of fluorine = 7
So, the total valence electron contribution = \[[8+(7\times 4)]\]= 36 electrons
The nearest multiple of noble gas is 32.
So, there is (36-32) = 4 that is 2 lone pairs of electrons on the xenon atom in the axial position. The hybridisation is\[s{{p}^{3}}{{d}^{2}}\]. The molecular shape is square planar.
So, the correct option is D.
Note: - The structures of any compound depend on the hybridisation and the number of lone pairs and bond pairs.
- Order of repulsion which determines the position of lone pairs around the central metal (either equatorial or axial):
- Lone pair-lone pair > Lone pair-bond pair > Bond pair-bond pair
Complete step by step answer:
\[S{{F}_{4}}\]:-
Valency of sulphur = 6
Valency of fluorine = 7
So, the total valence electron contribution = \[[6+(7\times 4)]\]= 34 electrons
The nearest multiple of noble gas is 32.
So, (34-32) = 2 electrons exist as a lone pair on the sulphur atom.
Now, the sulphur forms four single bonds to the fluorine atoms. Each fluorine atom has three lone pairs to complete their octet.
Since a lone pair experiences repulsion, it is placed in equatorial position to reduce repulsion. Sulphur forms four single bonds and one lone pair. So, the hybridisation is \[\text{s}{{\text{p}}^{3}}\text{d}\]. The molecular shape is trigonal bipyramidal.

\[C{{F}_{4}}\]:-
Valency of carbon = 4
Valency of fluorine = 7
So, the total valence electron contribution = \[[4+(7\times 4)]\]= 32 electrons
The nearest multiple of noble gas is 32.
So, there is no lone pair of electrons on the carbon atom. The hybridisation is \[\text{s}{{\text{p}}^{3}}\]. The molecular shape is tetrahedral.

\[Xe{{F}_{4}}\]:-
Valency of xenon = 8
Valency of fluorine = 7
So, the total valence electron contribution = \[[8+(7\times 4)]\]= 36 electrons
The nearest multiple of noble gas is 32.
So, there is (36-32) = 4 that is 2 lone pairs of electrons on the xenon atom in the axial position. The hybridisation is\[s{{p}^{3}}{{d}^{2}}\]. The molecular shape is square planar.

So, the correct option is D.
Note: - The structures of any compound depend on the hybridisation and the number of lone pairs and bond pairs.
- Order of repulsion which determines the position of lone pairs around the central metal (either equatorial or axial):
- Lone pair-lone pair > Lone pair-bond pair > Bond pair-bond pair
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




