The melting points of amino-acids are higher than the corresponding halo-acids because?
(a) Amino acids exist as zwitterion resulting in strong dipole-dipole interaction.
(b) Amino acids are optically active.
(c) Due to higher molecular mass of $-{ NH }_{ 2 }$ group molecular mass of amino acids is higher.
(d) They interact with water more than halo-acids and have salt-like structure.
Answer
260.4k+ views
Hint:In Amino acids, $ -{ NH }_{ 2 }$ (basic) and $ -COOH$ (acidic) groups are present as functional groups and they can be classified as $ \alpha -,\quad \beta -,\quad \gamma -,\quad \delta -,$ etc. depending upon the relative position of the $ -{ NH }_{ 2 }$ group with respect to the $ -COOH$ group .
Complete step by step answer:
There are a total of twenty six $\alpha -$amino acids that are found in proteins and they differ based on the nature of their side chain groups that determines the properties of the protein.
$\alpha -$amino acids possess the following properties:
-They are colourless, non-volatile crystalline solids.
-They melt with decomposition at very high temperatures.
-They are soluble in water but are insoluble in non-polar solvents.
-They exist as internal salt or zwitterion: Since amino acids contain both an acidic group (carboxyl group) and a basic group (amino group), they neutralise each other through the transfer of a proton from the carboxyl group to the amino group present in the molecule. The resultant ion is called a dipolar ion.
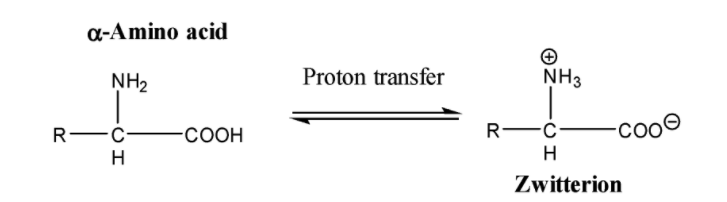
It is because of this zwitterion structure that the $\alpha -$amino acids have a crystalline structure and possess a high melting point.
Halo-acids are halogen derivatives of the carboxylic acids example 2-Chlorocarboxylic acids. Because they do comprise of halogen and –COOH groups, these molecules will possess dipole-dipole interactions but amino acids consist of stronger dipole-dipole interactions as well as electrostatic forces of attractions because of their dipolar nature.
Hence the correct answer is (a) Amino acids exist as zwitterion resulting in strong dipole-dipole interaction.
Note:
The chemical properties of the members of a homologous series similar though the first member may vary considerably from the rest of the members. The successive members of a homologous series differ by a \[C{{H}_{2}}\] group or by 14 mass units.
Complete step by step answer:
There are a total of twenty six $\alpha -$amino acids that are found in proteins and they differ based on the nature of their side chain groups that determines the properties of the protein.
$\alpha -$amino acids possess the following properties:
-They are colourless, non-volatile crystalline solids.
-They melt with decomposition at very high temperatures.
-They are soluble in water but are insoluble in non-polar solvents.
-They exist as internal salt or zwitterion: Since amino acids contain both an acidic group (carboxyl group) and a basic group (amino group), they neutralise each other through the transfer of a proton from the carboxyl group to the amino group present in the molecule. The resultant ion is called a dipolar ion.

It is because of this zwitterion structure that the $\alpha -$amino acids have a crystalline structure and possess a high melting point.
Halo-acids are halogen derivatives of the carboxylic acids example 2-Chlorocarboxylic acids. Because they do comprise of halogen and –COOH groups, these molecules will possess dipole-dipole interactions but amino acids consist of stronger dipole-dipole interactions as well as electrostatic forces of attractions because of their dipolar nature.
Hence the correct answer is (a) Amino acids exist as zwitterion resulting in strong dipole-dipole interaction.
Note:
The chemical properties of the members of a homologous series similar though the first member may vary considerably from the rest of the members. The successive members of a homologous series differ by a \[C{{H}_{2}}\] group or by 14 mass units.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




