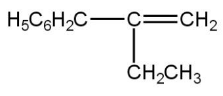
The major product of the following reaction is:
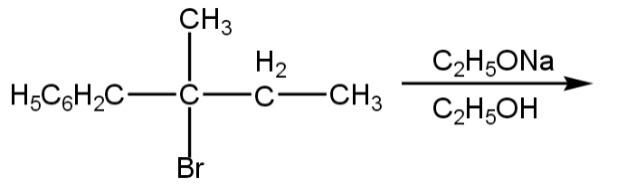
(A)
(B)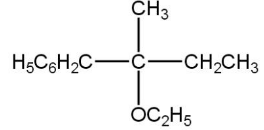
(C)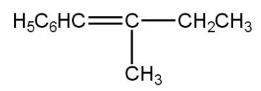
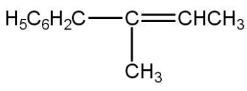
(D)
Answer
256.2k+ views
Hint: Given compound name is \[\left( 2-bromo-2-methyl\text{ }butyl \right)\] benzene and sodium ethoxide (\[{{C}_{2}}{{H}_{5}}ONa\]) which acts as a strong nucleophile reagent and also a strong base and thus, it tend to donate electron or seek cation. The reaction between both the compounds is followed by a substitution reaction (one nucleophile will substitute for another nucleophile).
Complete Step by Step Solution:
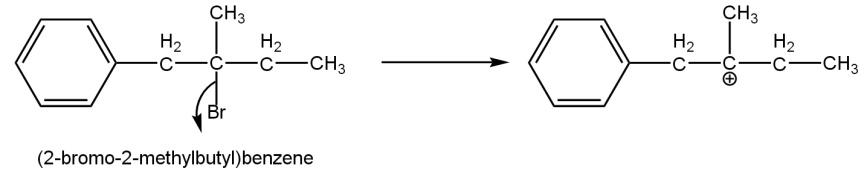
In a given compound \[\left( 2-bromo-2-methyl\text{ }butyl \right)\], bromine’s electronegativity is quite high as compared to carbon to which it is bonded. Due to its high electronegativity, it tends to attract a bond pair of electrons towards itself and become negatively charged. This results in creation of three-degree carbonation such as
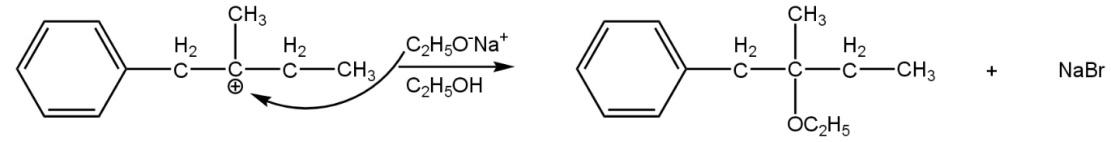
The resultant compound is unstable because it is a charge compound and not neutral compound and it tends to get neutral by the attack of another nucleophile. Sodium ethoxide (\[{{C}_{2}}{{H}_{5}}ONa\]) and ethanol (\[{{C}_{2}}{{H}_{5}}OH\]) are strong nucleophilic reagents (electron-rich), attack on it and side product will be\[NaBr\](attacked by sodium ethoxide) or\[HBr\] (attack by ethanol) such as
The major product is obtained by nucleophile substitution in this reaction and products obtained by elimination reaction are minor.
Thus, the correct option is A.
Note: Bromine (leaving group) is a good nucleophile due to which substitution reaction is preferred. If in the place of bromine, a strong and bulky base is present then an elimination reaction takes place and the major product will be obtained by elimination reaction and not by a substitution reaction. Also, neutral species are more stable than charged species.
Complete Step by Step Solution:
In a given compound \[\left( 2-bromo-2-methyl\text{ }butyl \right)\], bromine’s electronegativity is quite high as compared to carbon to which it is bonded. Due to its high electronegativity, it tends to attract a bond pair of electrons towards itself and become negatively charged. This results in creation of three-degree carbonation such as

The resultant compound is unstable because it is a charge compound and not neutral compound and it tends to get neutral by the attack of another nucleophile. Sodium ethoxide (\[{{C}_{2}}{{H}_{5}}ONa\]) and ethanol (\[{{C}_{2}}{{H}_{5}}OH\]) are strong nucleophilic reagents (electron-rich), attack on it and side product will be\[NaBr\](attacked by sodium ethoxide) or\[HBr\] (attack by ethanol) such as

The major product is obtained by nucleophile substitution in this reaction and products obtained by elimination reaction are minor.
Thus, the correct option is A.
Note: Bromine (leaving group) is a good nucleophile due to which substitution reaction is preferred. If in the place of bromine, a strong and bulky base is present then an elimination reaction takes place and the major product will be obtained by elimination reaction and not by a substitution reaction. Also, neutral species are more stable than charged species.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




