The formal charges of C and O atoms in $C{O_2}$ are respectively:
A. 1,-1
B. -1,1
C. 2,-1
D. 0,0
Answer
259.8k+ views
Hint: Chemical bonding refers to the formation of a chemical bond between two or more atoms, molecules, or ions to give rise to a chemical compound. These chemical bonds are what keeps the atoms together in the resulting compound. Moreover, formal charge is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity.
Complete step by step answer:
Formal charge is used to predict the most correct structure. Now, in the question we are given $C{O_2}$ molecule. Now, let’s draw all the resonating structures.
These are the resonating structures:
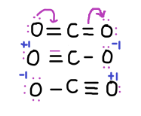
Now, $C{O_2}$ is a neutral molecule with 16 valence electrons. A valence electron is an outer shell electron that is associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed.
Further, let us consider all the three ways possible.
First, carbon single bonded to both oxygen atoms. (Carbon+2, oxygen-1 each, so total formal charge is 0)
Second, carbon single bonded to one oxygen and double bonded to another. (Carbon= $ + 1$, oxygen double=0, oxygen single= -1, so total formal charge is 0)
Third, carbon double bonded to both oxygen atoms (carbon=0, oxygen=0 so formal charge=0
Therefore, the formal charge of the C and O atom in $C{O_2}$ molecule is zero.
Hence, option D is correct.
Note:
Carbon dioxide becomes a poisonous gas when inhaled in large amounts. It can lead to many respiratory disorders and can cause severe damage to the nervous system. $C{O_2}$ in the form of liquid and solid is used for refrigeration and cooling. It is used as an inert gas in various chemical processes. It is also used in the manufacture of casting molds in order to enhance their hardness.
Complete step by step answer:
Formal charge is used to predict the most correct structure. Now, in the question we are given $C{O_2}$ molecule. Now, let’s draw all the resonating structures.
These are the resonating structures:
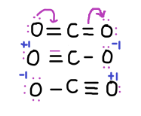
Now, $C{O_2}$ is a neutral molecule with 16 valence electrons. A valence electron is an outer shell electron that is associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed.
Further, let us consider all the three ways possible.
First, carbon single bonded to both oxygen atoms. (Carbon+2, oxygen-1 each, so total formal charge is 0)
Second, carbon single bonded to one oxygen and double bonded to another. (Carbon= $ + 1$, oxygen double=0, oxygen single= -1, so total formal charge is 0)
Third, carbon double bonded to both oxygen atoms (carbon=0, oxygen=0 so formal charge=0
Therefore, the formal charge of the C and O atom in $C{O_2}$ molecule is zero.
Hence, option D is correct.
Note:
Carbon dioxide becomes a poisonous gas when inhaled in large amounts. It can lead to many respiratory disorders and can cause severe damage to the nervous system. $C{O_2}$ in the form of liquid and solid is used for refrigeration and cooling. It is used as an inert gas in various chemical processes. It is also used in the manufacture of casting molds in order to enhance their hardness.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




