The difference between amylase and amylopectin is __________.
(a)- \[Amylopectin\text{ }have\text{ }1\to 4\alpha -linkage\text{ }and\text{ }1\to 6\beta -linkage\]
(b)- \[Amylopectin\text{ }have\text{ }1\to 4\alpha -linkage\text{ }and\text{ }1\to 6\alpha -linkage\]
(c)- Amylose is made up of glucose and galactose.
(d)- \[Amylose\text{ }have\text{ }1\to 4\alpha -linkage\text{ }and\text{ }1\to 6\beta -linkage\]
Answer
264.6k+ views
Hint: One of the following has the branched-chain structure and one has the linear chain structure. Both of them are made up of glucose components only.
Complete step by step answer:
Starch is not only a single compound but is a mixture of two components-a water-soluble components called amylose (15-20%) and a water-insoluble component called amylopectin (80-85%). The aqueous solution of amylose gives a blue color with an iodine solution. Amylopectin, on the other hand, does not give blue color with iodine solution.
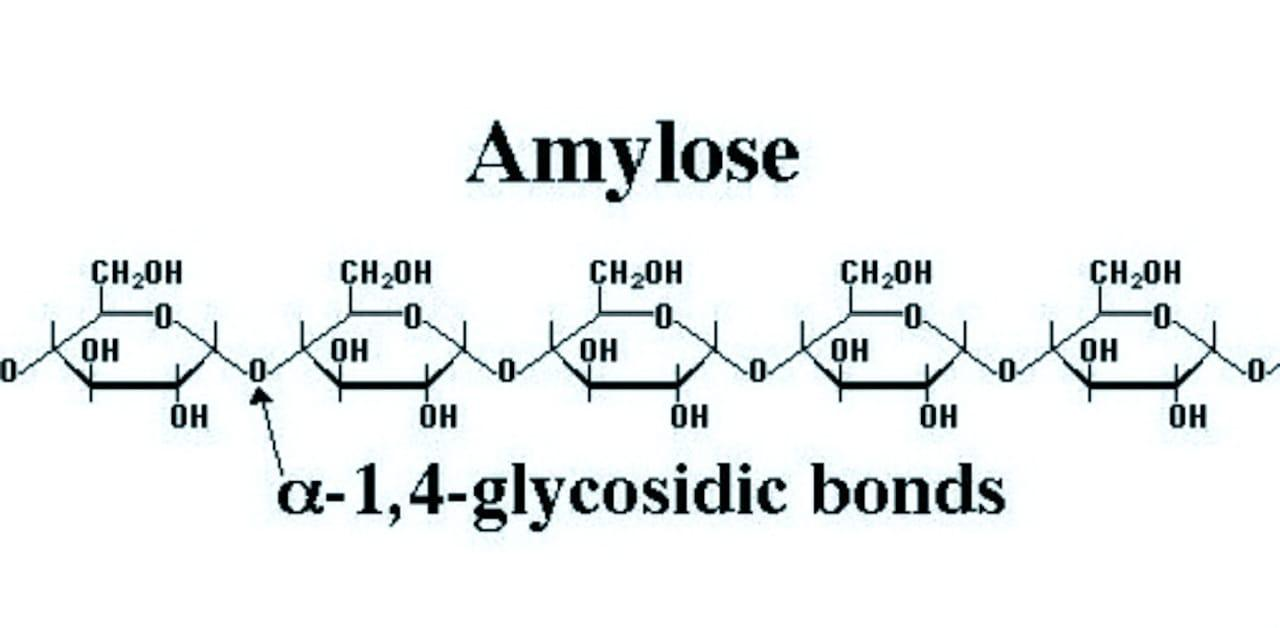
Structure of amylose: It is a linear polymer of \[\alpha -D-glu\cos e\] in which\[{{C}_{1}}\] of one glucose unit is attached to\[{{C}_{4}}\] of the other through\[\alpha -gly\cos idic\] linkage.
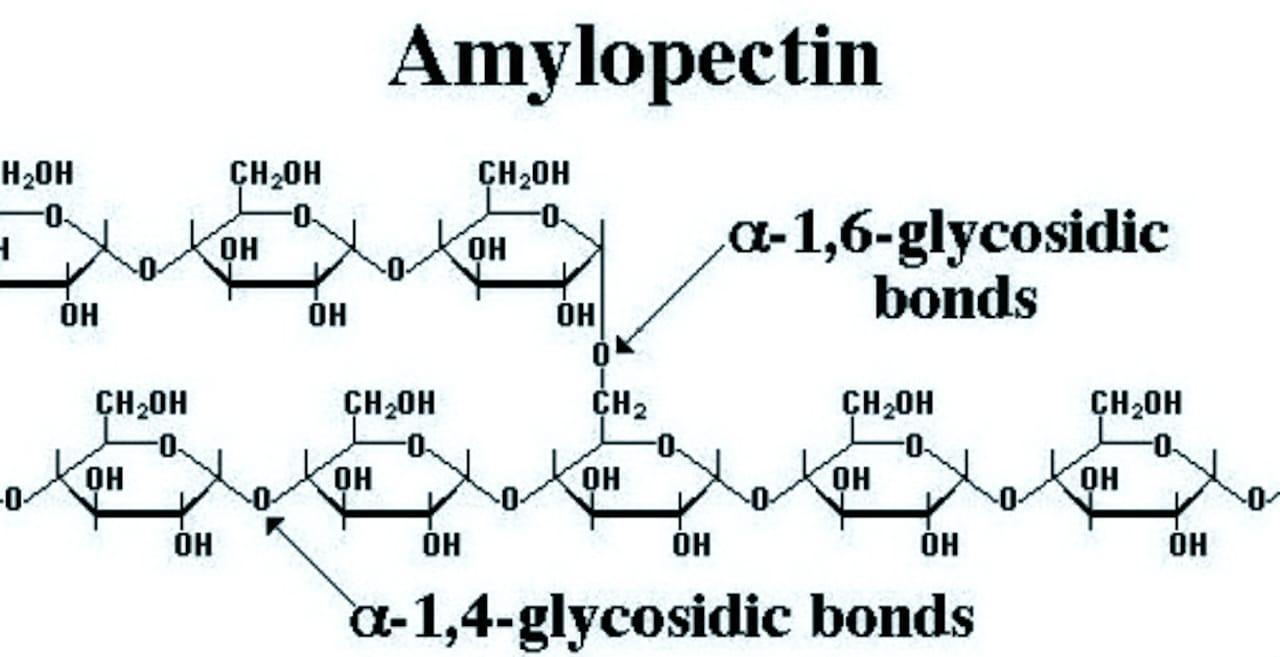
Structure of amylopectin: Amylopectin, on the other hand, is a highly branched polymer. It consists of a large number of short chains each containing 20-25 glucose units which are joined together through \[\alpha -gly\cos idic\] linkage involving \[{{C}_{1}}\] of one glucose unit with\[{{C}_{4}}\] of the other. The \[{{C}_{1}}\]of the terminal glucose unit in each chain is further linked to\[{{C}_{6}}\] of some other glucose unit in the next chain through \[{{C}_{1}}-{{C}_{6}}\]\[\alpha -gly\cos idic\] linkage. This gives amylopectin a highly branched structure.
Hence, from the above discussion option (b) is correct.
Note: You may get confused between \[\alpha -linkage\] and \[\beta -linkage\]. \[\alpha -linkage\] is formed when the bond is formed on the same side and \[\beta -linkage\]is formed when the bond is formed between the opposite side.
Complete step by step answer:
Starch is not only a single compound but is a mixture of two components-a water-soluble components called amylose (15-20%) and a water-insoluble component called amylopectin (80-85%). The aqueous solution of amylose gives a blue color with an iodine solution. Amylopectin, on the other hand, does not give blue color with iodine solution.
Structure of amylose: It is a linear polymer of \[\alpha -D-glu\cos e\] in which\[{{C}_{1}}\] of one glucose unit is attached to\[{{C}_{4}}\] of the other through\[\alpha -gly\cos idic\] linkage.

Structure of amylopectin: Amylopectin, on the other hand, is a highly branched polymer. It consists of a large number of short chains each containing 20-25 glucose units which are joined together through \[\alpha -gly\cos idic\] linkage involving \[{{C}_{1}}\] of one glucose unit with\[{{C}_{4}}\] of the other. The \[{{C}_{1}}\]of the terminal glucose unit in each chain is further linked to\[{{C}_{6}}\] of some other glucose unit in the next chain through \[{{C}_{1}}-{{C}_{6}}\]\[\alpha -gly\cos idic\] linkage. This gives amylopectin a highly branched structure.

Hence, from the above discussion option (b) is correct.
Note: You may get confused between \[\alpha -linkage\] and \[\beta -linkage\]. \[\alpha -linkage\] is formed when the bond is formed on the same side and \[\beta -linkage\]is formed when the bond is formed between the opposite side.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




