The basicity of
(I) o—toluidine
(II) m—toluidine
(III) p—toluidine
(IV) aniline
follow the order:
A. I>II>III>IV
B. IV>III>II>I
C. III>II>IV>I
D. I>IV>II>III
Answer
266.4k+ views
Hint: According to the concept of Lewis acid-base theory the compound or molecule can accept hydrogen ion is known as base. Higher the tendency to accept hydrogen ion higher will be the basicity of that compound.
Complete step by step solution:
Toluidine is an aromatic amine. The basicity of the aromatic amine is depending upon the availability of the lone pair. Higher the lone pair availability higher the donating ability of the lone pair as well as accept tendency of hydrogen ions.
In case of aromatic amine, the lone pair undergoes conjugation with the benzene . As a result, lone pair availability decreases. Therefore, in presence of an electron donating group increases the tendency of lone pair availability as well as basicity.
The structures of o—toluidine, m—toluidine, p—toluidine, aniline as follows,
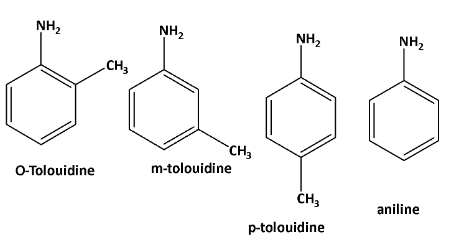
Now \[{\text{ - C}}{{\text{H}}_{\text{3}}}\] is an electron donating group. due to the positive inductive effect of the methyl group the lone pair of amine availability increases as well as the basicity.
In the case of o—toluidine, due to the steric hindrance of the methyl group , after accepting the hydrogen ion the ammonium ion gets out of the plane. As a result, corresponding conjugate acid stability gets decreased. That is why the basicity of o—toluidine is least among others.
In the case of p—toluidine due the hyperconjugation of methyl groups the basicity increases greater than others.
So, the overall order is, p—toluidine> m—toluidine> aniline> o—toluidine,
So, the correct option is, C. III>II>IV>I
Note: Acidity of an organic compound is depending upon the electron deficiency of the hydrogen atom. Higher the electron deficiency of the hydrogen higher will be the acidity character of that hydrogen.
Now to be an acidic hydrogen that hydrogen should be attached with a high electronegative group or electron withdrawing group. Higher the electronegativity of the group, higher will be the electron deficiency of the hydrogen, attached with that group as well as the acidity.
Complete step by step solution:
Toluidine is an aromatic amine. The basicity of the aromatic amine is depending upon the availability of the lone pair. Higher the lone pair availability higher the donating ability of the lone pair as well as accept tendency of hydrogen ions.
In case of aromatic amine, the lone pair undergoes conjugation with the benzene . As a result, lone pair availability decreases. Therefore, in presence of an electron donating group increases the tendency of lone pair availability as well as basicity.
The structures of o—toluidine, m—toluidine, p—toluidine, aniline as follows,

Now \[{\text{ - C}}{{\text{H}}_{\text{3}}}\] is an electron donating group. due to the positive inductive effect of the methyl group the lone pair of amine availability increases as well as the basicity.
In the case of o—toluidine, due to the steric hindrance of the methyl group , after accepting the hydrogen ion the ammonium ion gets out of the plane. As a result, corresponding conjugate acid stability gets decreased. That is why the basicity of o—toluidine is least among others.
In the case of p—toluidine due the hyperconjugation of methyl groups the basicity increases greater than others.
So, the overall order is, p—toluidine> m—toluidine> aniline> o—toluidine,
So, the correct option is, C. III>II>IV>I
Note: Acidity of an organic compound is depending upon the electron deficiency of the hydrogen atom. Higher the electron deficiency of the hydrogen higher will be the acidity character of that hydrogen.
Now to be an acidic hydrogen that hydrogen should be attached with a high electronegative group or electron withdrawing group. Higher the electronegativity of the group, higher will be the electron deficiency of the hydrogen, attached with that group as well as the acidity.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




