What is the structure of $$XeO{F_4}$$ ?
(A) Square pyramidal
(B) Trigonal bipyramidal
(C) Pyramidal
(D) Square bipyramidal
Answer
266.4k+ views
Hint: With the help of lewis structure, we can know the bonding in the compound and predict the shape. Xenon is the central atom in this structure. Hybridization in this molecule is $$s{p^3}{d^2}$$.
Complete Step-by-Step Answer:
We can draw the lewis structure of the compound, so that we will get the idea about the shape of the molecule.
We know that Fluorine generally binds with one atom and oxygen generally binds with two atoms. This structure involves six atoms. So, Xenon must be the central atom.
In the lewis structure, Xenon has 8 valence electrons as it is a noble gas, oxygen has 6 electrons and fluorine has 7 valence electrons. Let’s make bonds between them and see the structure.
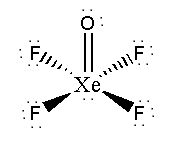
We can see that xenon uses 4 of its electrons to make bonds with 4 fluorine atoms. 2 of the valence electrons of xenon make double bonds with oxygen and the remaining two are as a lone pair.
As lone pairs have strong repulsive effects on bonds, Xe-F bonds have a tendency to be as far as possible from the lone pair.
Xenon-Oxygen double bonds also have a repulsive effect as oxygen and fluorine atoms have 2 and 3 lone pairs respectively.
So, Xe-F bonds lie in a plane to make repulsive forces least as possible and on two sides of the plane, oxygen atoms and lone pairs reside.
So, the correct answer is (A) Square pyramidal.
Additional Information:
VSEPR theory is also helpful in order to determine the geometry of the molecule. VSEPR stands for Valence shell electron pair repulsion theory. It involves consideration of various electron pairs present in the molecule and considering those repulsive forces, structure is assigned.
Note: As $$XeO{F_4}$$ has 5 other atoms than the central atom xenon, it simply doesn’t mean that it has trigonal bipyramidal structure. Do not forget to consider the lone pair in the central atom as well because it has the highest repulsive forces on the atoms which are bonded with the central atom.
Complete Step-by-Step Answer:
We can draw the lewis structure of the compound, so that we will get the idea about the shape of the molecule.
We know that Fluorine generally binds with one atom and oxygen generally binds with two atoms. This structure involves six atoms. So, Xenon must be the central atom.
In the lewis structure, Xenon has 8 valence electrons as it is a noble gas, oxygen has 6 electrons and fluorine has 7 valence electrons. Let’s make bonds between them and see the structure.

We can see that xenon uses 4 of its electrons to make bonds with 4 fluorine atoms. 2 of the valence electrons of xenon make double bonds with oxygen and the remaining two are as a lone pair.
As lone pairs have strong repulsive effects on bonds, Xe-F bonds have a tendency to be as far as possible from the lone pair.
Xenon-Oxygen double bonds also have a repulsive effect as oxygen and fluorine atoms have 2 and 3 lone pairs respectively.
So, Xe-F bonds lie in a plane to make repulsive forces least as possible and on two sides of the plane, oxygen atoms and lone pairs reside.
So, the correct answer is (A) Square pyramidal.
Additional Information:
VSEPR theory is also helpful in order to determine the geometry of the molecule. VSEPR stands for Valence shell electron pair repulsion theory. It involves consideration of various electron pairs present in the molecule and considering those repulsive forces, structure is assigned.
Note: As $$XeO{F_4}$$ has 5 other atoms than the central atom xenon, it simply doesn’t mean that it has trigonal bipyramidal structure. Do not forget to consider the lone pair in the central atom as well because it has the highest repulsive forces on the atoms which are bonded with the central atom.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




