Structure formula of \[{H_2}{O_2}\] is
(a) 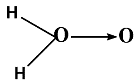
(b) \[H - O - O - H\](Straight line).
(c) Where, \[\angle H - O - O = \angle O - O - H' = {101.5^o}\]and all the four atoms are in the same plane. 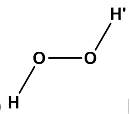
(d) Where, \[\angle H - O - O = \angle O - O - H' = {97^o}\]and the angle between \[H - O - O\]plane and \[O - O - H'\]plane is \[{101^o}\]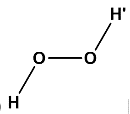
Answer
267.3k+ views
Hint: Hydrogen peroxide (\[{H_2}{O_2}\]) is the type of simplest known peroxide with oxygen-oxygen single bond. It has a non-planar and open book-type structure. Depending upon the reaction medium, it can behave like an oxidizing and reducing agent.
Complete step by step solution:Hydrogen peroxide is like water with an extra oxygen atom. Due to the presence of \[ - OH\] groups, it has an acidic nature( \[pH = 4.5\]).
Hydrogen peroxide is \[100\% \]degradable compound.
Hydrogen peroxide is considered as non-planar in nature with an open book-like structure.
The structure of hydrogen peroxide indicates the presence of an oxygen-oxygen single bond( \[O - O\]) with bond length \[145.5pm\]. Whereas the \[O - H\]bond length is around \[98.8pm\].
The non-planar open-book structure of hydrogen peroxide has two planes and both the planes contain \[OH\]group. The bond angle between both the planes is \[{90.2^o}\].
The bond angle between the \[H - O - O\]is \[101.{9^o}\].
The open-book structure of hydrogen peroxide can be represented as below.
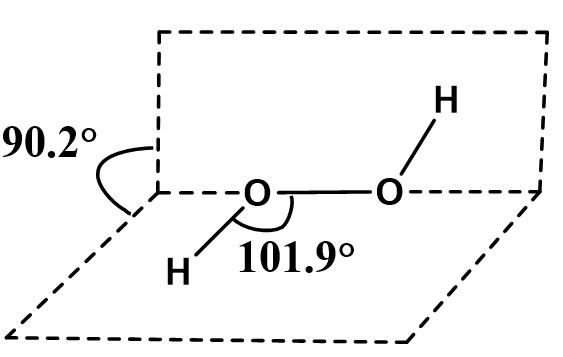
Image: An open-book structure of hydrogen peroxide.
Therefore from the above explanation we can say option (d) will be the correct option:
Note: The purest form of hydrogen peroxide is colorless in nature.
Hydrogen peroxide melts at \[272.4K\]and boils at \[423K\].
Hydrogen peroxide is miscible in water in all ratios.
The oxygen atom has -1 oxidation state in hydrogen peroxide.
Hydrogen peroxide is also used as a bleaching agent.
Complete step by step solution:Hydrogen peroxide is like water with an extra oxygen atom. Due to the presence of \[ - OH\] groups, it has an acidic nature( \[pH = 4.5\]).
Hydrogen peroxide is \[100\% \]degradable compound.
Hydrogen peroxide is considered as non-planar in nature with an open book-like structure.
The structure of hydrogen peroxide indicates the presence of an oxygen-oxygen single bond( \[O - O\]) with bond length \[145.5pm\]. Whereas the \[O - H\]bond length is around \[98.8pm\].
The non-planar open-book structure of hydrogen peroxide has two planes and both the planes contain \[OH\]group. The bond angle between both the planes is \[{90.2^o}\].
The bond angle between the \[H - O - O\]is \[101.{9^o}\].
The open-book structure of hydrogen peroxide can be represented as below.

Image: An open-book structure of hydrogen peroxide.
Therefore from the above explanation we can say option (d) will be the correct option:
Note: The purest form of hydrogen peroxide is colorless in nature.
Hydrogen peroxide melts at \[272.4K\]and boils at \[423K\].
Hydrogen peroxide is miscible in water in all ratios.
The oxygen atom has -1 oxidation state in hydrogen peroxide.
Hydrogen peroxide is also used as a bleaching agent.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




