State the electronic configuration for Neon $\left[ {P = 10,n = 10} \right]$
A. $2,{\text{ 6}}$
B. $2,{\text{ 7}}$
C. $2,{\text{ 9}}$
D. $2,{\text{ 8}}$
Answer
262.8k+ views
Hint: Electronic configuration is the distribution of electrons into different shells, subshells and orbitals in an atom. The electronic configuration is governed by Aufbau’s principle and some other rules.
Complete step by step solution:
Aufbau’s principle helps in filling of electrons in various shells and subshells according to the energy of electrons a representation of the rule is drawn as,
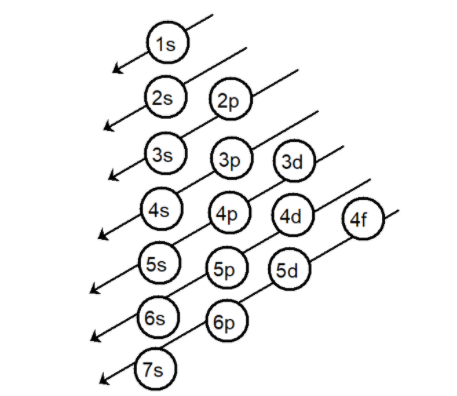
The number of protons is equal to the number of electrons hence the element has $10$ electrons. According to Aufbau’s principle the electrons are filled as,
Electronic configuration of element with atomic number $10$is,
\[{}_{10}Ne = \]$1{s^2}2{s^2}2{p^6}$
From the above given information, we can calculate the electronic configuration of Neon.
The symbol of Neon is $Ne$, and the atomic number of Neon is $10$. Then the electronic configuration of Neon is $1{s^2}2{s^2}2{p^6}$
The inner shell $\left( {n = 1} \right)$contains two electrons and outer shell $\left( {n = 2} \right)$contains eight electrons. Therefore, electronic configuration of Neon is$2,{\text{ 8}}$.
Hence, the correct option is (D).
Addition information:
(i) Electronic configuration of the element is written in terms of ${s^x},{\text{ }}{{\text{p}}^y},{\text{ }}{{\text{d}}^z}$. In this type of notation, the subshells are represented by their respective symbol for example, $s$is written for the subshell with $l = 0$. $p$is written for subshell with $l = 1.$ In the notation, the letters $x,{\text{ y, z}}$….. represent the number of electrons present in the various subshells. For the complete electronic configuration, the principal quantum number is written before the respective subshell. For example: electronic configuration of $Mg\left( {12} \right) = 1{s^2}2{s^2}s{p^6}3{s^2}$
(ii) Orbital notation:
It is the other way of expressing the electronic configuration of an atom. In this notation, the orbitals of the subshell are represented by a box and electrons are represented by the arrows. Arrow $\left( \uparrow \right)$represents $ + \dfrac{1}{2}$i.e. positive spin and an arrow $\left( \downarrow \right)$represents$ - \dfrac{1}{2}$ i.e. negative spin.
For example: magnesium

Note: Aufbau’s principle: this principle states that the electrons are added one by one into the various orbitals in order of their increasing energy starting with the orbital of lowest energy. In other words, we can say that the electrons first occupy the lowest energy orbital available to them and only when these orbitals are filled, the only the electrons are filled in the higher energy orbitals. For multi-electron atoms, the following is the increasing order of the energy of the orbitals are 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s. For the prediction of orbitals, only three quantum numbers are required [$n,{\text{ l, m}}$] and for the prediction of electrons all quantum numbers are required [$n,{\text{ l, m}}$ and $s$].
Complete step by step solution:
Aufbau’s principle helps in filling of electrons in various shells and subshells according to the energy of electrons a representation of the rule is drawn as,

The number of protons is equal to the number of electrons hence the element has $10$ electrons. According to Aufbau’s principle the electrons are filled as,
Electronic configuration of element with atomic number $10$is,
\[{}_{10}Ne = \]$1{s^2}2{s^2}2{p^6}$
From the above given information, we can calculate the electronic configuration of Neon.
The symbol of Neon is $Ne$, and the atomic number of Neon is $10$. Then the electronic configuration of Neon is $1{s^2}2{s^2}2{p^6}$
The inner shell $\left( {n = 1} \right)$contains two electrons and outer shell $\left( {n = 2} \right)$contains eight electrons. Therefore, electronic configuration of Neon is$2,{\text{ 8}}$.
Hence, the correct option is (D).
Addition information:
(i) Electronic configuration of the element is written in terms of ${s^x},{\text{ }}{{\text{p}}^y},{\text{ }}{{\text{d}}^z}$. In this type of notation, the subshells are represented by their respective symbol for example, $s$is written for the subshell with $l = 0$. $p$is written for subshell with $l = 1.$ In the notation, the letters $x,{\text{ y, z}}$….. represent the number of electrons present in the various subshells. For the complete electronic configuration, the principal quantum number is written before the respective subshell. For example: electronic configuration of $Mg\left( {12} \right) = 1{s^2}2{s^2}s{p^6}3{s^2}$
(ii) Orbital notation:

It is the other way of expressing the electronic configuration of an atom. In this notation, the orbitals of the subshell are represented by a box and electrons are represented by the arrows. Arrow $\left( \uparrow \right)$represents $ + \dfrac{1}{2}$i.e. positive spin and an arrow $\left( \downarrow \right)$represents$ - \dfrac{1}{2}$ i.e. negative spin.
For example: magnesium

Note: Aufbau’s principle: this principle states that the electrons are added one by one into the various orbitals in order of their increasing energy starting with the orbital of lowest energy. In other words, we can say that the electrons first occupy the lowest energy orbital available to them and only when these orbitals are filled, the only the electrons are filled in the higher energy orbitals. For multi-electron atoms, the following is the increasing order of the energy of the orbitals are 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s. For the prediction of orbitals, only three quantum numbers are required [$n,{\text{ l, m}}$] and for the prediction of electrons all quantum numbers are required [$n,{\text{ l, m}}$ and $s$].
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




