Select correct statement for $Br{F_5}.$
(A) All fluorine atoms are in the same plane.
(B) Four fluorine atoms and Br atoms are in the same plane.
(C) Four fluorine atoms are in the same plane.
(D) It has\[F - Br - F\]bond angles at ${90^\circ }.$
Answer
263.7k+ views
Hint: $Br{F_5}$ is an interhalogen compound. When two different halogens form compounds among themselves, interhalogen compounds are formed. Such compound may have general formula $X{X^1},XX_3^1,XX_5^1$ and $XX_7^1.$ Here $X$ is halogen of large size and ${X^1}$of small size and $X$ is more negative.
Complete step by step answer:
In $Br{F_5}$Bromine is the central atom and fluorine atoms present around it.
The name of the compound is Bromine pentafluoride.
Bromine atoms have larger size than fluorine and fluorine is more electronegative.
Electronic configuration of Br, $1{s^2}2{s^2}2{p^6}2{s^2}3{p^6}3{d^{10}}4{s^2}4p_x^24p_y^24p_z^1$
Ground state configuration =
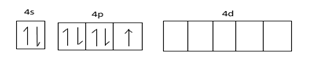
Excited state configuration =
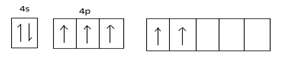
Hybridized state =
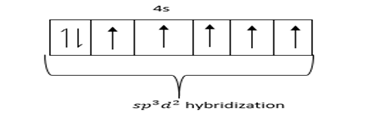
Geometry of $Br{F_5}$ is square pyramidal and hybridization $s{p^3}{d^2}.$
In $s{p^3}{d^2},$ 4 Fluorine atoms and one Br-atom present in the same plane.
Bond angle between them is 90 Degrees.
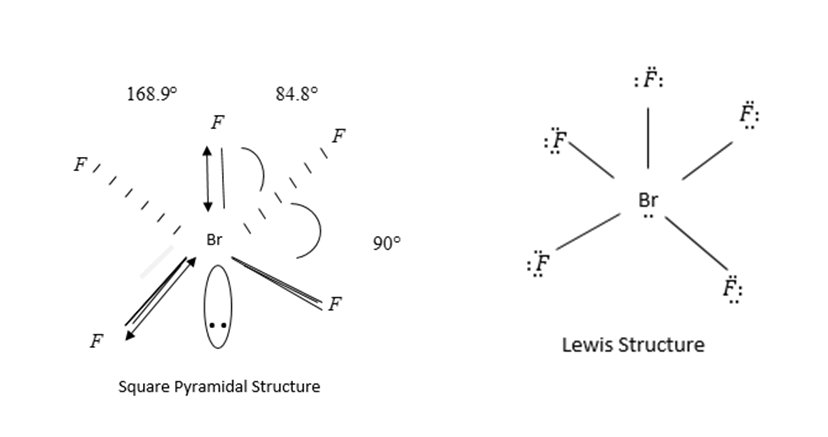
In Lewis structure we see that octets of each element should be complete. On fluorine atoms 3 lone pairs of electrons and on bromine atom only one lone pair is present.
By VSEPR theory 4 bond pairs occupy corners of squares and one bond pair and lone pair of electrons occupy equatorial position to minimize repulsion between lone pair and bond pair of electron.
Interhalogen compounds are covalent in nature due to low electronegativity difference between halogens. They are diamagnetic in nature. They are more reactive than halogen.
Interhalogen compounds behave as strong oxidizing agents and attack other elements to give a mixture of halides.
Therefore, the above explanation, the correct option is [D] Both assertion and reason are incorrect.
Note: Interhalogen compounds are more reactive than halogen. This is because the $X - {X^ - }$ bond present in them is weaker than $X - X$ and${X^1} - X$ bonds.
Complete step by step answer:
In $Br{F_5}$Bromine is the central atom and fluorine atoms present around it.
The name of the compound is Bromine pentafluoride.
Bromine atoms have larger size than fluorine and fluorine is more electronegative.
Electronic configuration of Br, $1{s^2}2{s^2}2{p^6}2{s^2}3{p^6}3{d^{10}}4{s^2}4p_x^24p_y^24p_z^1$
Ground state configuration =

Excited state configuration =

Hybridized state =

Geometry of $Br{F_5}$ is square pyramidal and hybridization $s{p^3}{d^2}.$
In $s{p^3}{d^2},$ 4 Fluorine atoms and one Br-atom present in the same plane.
Bond angle between them is 90 Degrees.

In Lewis structure we see that octets of each element should be complete. On fluorine atoms 3 lone pairs of electrons and on bromine atom only one lone pair is present.
By VSEPR theory 4 bond pairs occupy corners of squares and one bond pair and lone pair of electrons occupy equatorial position to minimize repulsion between lone pair and bond pair of electron.
Interhalogen compounds are covalent in nature due to low electronegativity difference between halogens. They are diamagnetic in nature. They are more reactive than halogen.
Interhalogen compounds behave as strong oxidizing agents and attack other elements to give a mixture of halides.
Therefore, the above explanation, the correct option is [D] Both assertion and reason are incorrect.
Note: Interhalogen compounds are more reactive than halogen. This is because the $X - {X^ - }$ bond present in them is weaker than $X - X$ and${X^1} - X$ bonds.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding Electromagnetic Waves and Their Importance




