$Sb{{F}_{5}}$ reacts with $Xe{{F}_{4}}$ and $Xe{{F}_{6}}$ to form ionic compounds $\left[ XeF_{3}^{+} \right]\left[ SbF_{6}^{-} \right]$ and $\left[ XeF_{5}^{+} \right]\left[ SbF_{6}^{-} \right]$. The geometry of $XeF_{3}^{+}$, $\left[ XeF_{5}^{+} \right]$ ion respectively is:
(A) square pyramidal, T-shaped
(B) bent T-shaped, square pyramidal
(C) see-saw, square pyramidal
(D) square pyramidal, see-saw
Answer
259.8k+ views
Hint: By obtaining the hybridisation of the central metal atom with respect to the ligands by considering the oxidation state and the configuration of the central atom. Then, from the lone pairs and the bond pairs, the geometry can be determined.
Complete step by step solution:
It is given that the $Sb{{F}_{5}}$ reacts with $Xe{{F}_{4}}$ and $Xe{{F}_{6}}$, acting as a Lewis acid as follows :
\[Xe{{F}_{4}}+Sb{{F}_{5}}\to {{\left[ Xe{{F}_{3}} \right]}^{+}}{{\left[ Sb{{F}_{6}} \right]}^{-}}\]
$Xe{{F}_{6}}+Sb{{F}_{5}}\to {{\left[ Xe{{F}_{5}} \right]}^{+}}{{\left[ Sb{{F}_{6}} \right]}^{-}}$
Now, in order to find the geometry of the cationic sphere, we will use the VSEPR theory as follows:
-In both the cations, xenon being least electronegative, is the central atom. Its electronic configuration at ground state is $\left[ Kr \right]4{{d}^{10}}5{{s}^{2}}5{{p}^{6}}$.
-Due to the presence of a $(+1)$ charge on the sphere, that is, loss of an electron, the xenon loses one of its electrons from the 5p orbital. So, the configuration of $X{{e}^{+}}$ becomes $\left[ Kr \right]4{{d}^{10}}5{{s}^{2}}5{{p}^{5}}$.
-In case of $XeF_{3}^{+}$, in order to form three bonds with the fluorine atom. The xenon atom goes into an excited state and one of its 5p electrons jumps to 5d orbital. Now, these 5s, 5p and 5d-orbitals undergo hybridisation to form five $s{{p}^{3}}d$ hybrid orbitals, consisting of two lone pairs and three unpaired electrons.
-These unpaired electrons in the three hybrid orbitals overlap with an unpaired electron present in the three-fluorine p-orbital, one in each.
Thus, the hybridisation obtained in order to form $XeF_{3}^{+}$is $s{{p}^{3}}d$ hybridisation. So, the geometry will be trigonal bipyramidal, with one bond pair and two lone pairs in the equatorial position and the other two bond pairs in the axial position.
Due to the repulsion from the two lone pairs, we get a bent T-shaped molecule.
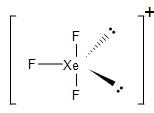
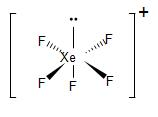
Similarly, in the case of $XeF_{5}^{+}$, in order to form five bond pairs with the fluorine atom. In the excited state, two p-electrons jump to the d-orbital. the 5s, 5p and 5d orbitals undergo hybridisation to form six $s{{p}^{3}}{{d}^{2}}$ hybrid orbitals. Thus, generating one lone pair and five unpaired electrons.
The five unpaired electrons overlap with the fluorine p-orbitals and form five bond pairs. Therefore, we get $s{{p}^{3}}{{d}^{2}}$ hybridisation and giving us an octahedral geometry. Due to the presence of the lone pair, its geometry gets distorted and we get a square pyramidal molecule.
Therefore, the shape of the $XeF_{3}^{+}$and $XeF_{5}^{+}$ entity is option (B)- bent T-shaped, square pyramidal.
Note: In order to obtain the hybridisation, the charge on the ion must be taken to obtain the oxidation state of the metal atom and then we would arrive at the appropriate hybridisation.
Complete step by step solution:
It is given that the $Sb{{F}_{5}}$ reacts with $Xe{{F}_{4}}$ and $Xe{{F}_{6}}$, acting as a Lewis acid as follows :
\[Xe{{F}_{4}}+Sb{{F}_{5}}\to {{\left[ Xe{{F}_{3}} \right]}^{+}}{{\left[ Sb{{F}_{6}} \right]}^{-}}\]
$Xe{{F}_{6}}+Sb{{F}_{5}}\to {{\left[ Xe{{F}_{5}} \right]}^{+}}{{\left[ Sb{{F}_{6}} \right]}^{-}}$
Now, in order to find the geometry of the cationic sphere, we will use the VSEPR theory as follows:
-In both the cations, xenon being least electronegative, is the central atom. Its electronic configuration at ground state is $\left[ Kr \right]4{{d}^{10}}5{{s}^{2}}5{{p}^{6}}$.
-Due to the presence of a $(+1)$ charge on the sphere, that is, loss of an electron, the xenon loses one of its electrons from the 5p orbital. So, the configuration of $X{{e}^{+}}$ becomes $\left[ Kr \right]4{{d}^{10}}5{{s}^{2}}5{{p}^{5}}$.
-In case of $XeF_{3}^{+}$, in order to form three bonds with the fluorine atom. The xenon atom goes into an excited state and one of its 5p electrons jumps to 5d orbital. Now, these 5s, 5p and 5d-orbitals undergo hybridisation to form five $s{{p}^{3}}d$ hybrid orbitals, consisting of two lone pairs and three unpaired electrons.
-These unpaired electrons in the three hybrid orbitals overlap with an unpaired electron present in the three-fluorine p-orbital, one in each.
Thus, the hybridisation obtained in order to form $XeF_{3}^{+}$is $s{{p}^{3}}d$ hybridisation. So, the geometry will be trigonal bipyramidal, with one bond pair and two lone pairs in the equatorial position and the other two bond pairs in the axial position.
Due to the repulsion from the two lone pairs, we get a bent T-shaped molecule.


Similarly, in the case of $XeF_{5}^{+}$, in order to form five bond pairs with the fluorine atom. In the excited state, two p-electrons jump to the d-orbital. the 5s, 5p and 5d orbitals undergo hybridisation to form six $s{{p}^{3}}{{d}^{2}}$ hybrid orbitals. Thus, generating one lone pair and five unpaired electrons.
The five unpaired electrons overlap with the fluorine p-orbitals and form five bond pairs. Therefore, we get $s{{p}^{3}}{{d}^{2}}$ hybridisation and giving us an octahedral geometry. Due to the presence of the lone pair, its geometry gets distorted and we get a square pyramidal molecule.
Therefore, the shape of the $XeF_{3}^{+}$and $XeF_{5}^{+}$ entity is option (B)- bent T-shaped, square pyramidal.
Note: In order to obtain the hybridisation, the charge on the ion must be taken to obtain the oxidation state of the metal atom and then we would arrive at the appropriate hybridisation.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




