Salicylic acid is prepared from phenol by:
A. Reimer-Tiemann reaction
B. Kolbe's reaction
C. Kolbe-electrolysis reaction
D. None of the above
Answer
268.8k+ views
Hint: Reaction of phenol with carbon tetrachloride in aqueous sodium hydroxide gives salicylic acid as the major product.
It is called the Reimer-Tiemann reaction.
Complete step-by-step answer:
In the reaction of phenol with carbon tetrachloride in aqueous sodium or potassium hydroxide, salicylic acid is the product.
This reaction occurs at 340 K temperature.
The IUPAC name of Salicylic acid is 2-hydroxybenzoic acid.
The mechanism of the reaction is as follows:
It is an electrophilic substitution reaction.
The first step involves the removal of the proton from phenol in aqueous sodium hydroxide giving sodium phenoxide as the product.
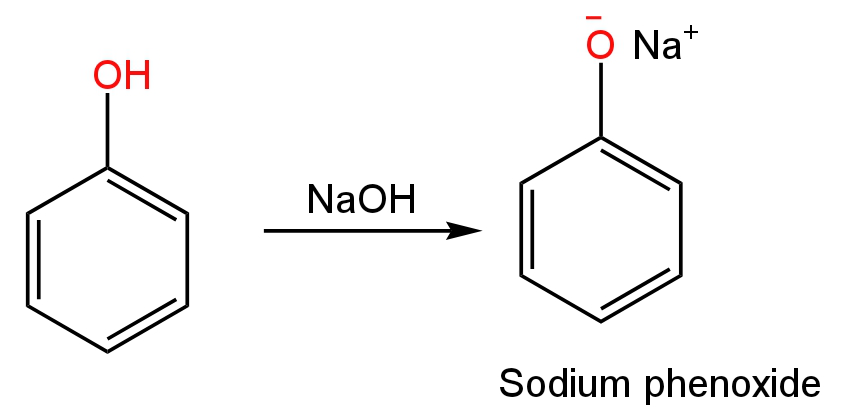
Image: Sodium phenoxide ion formation
The next step involves the attack of carbon tetrachloride at the ortho position of the benzene ring.
This leads to the replacement of three chlorine atoms with three hydroxide ions.
The removal of a molecule of water happens in the following step.
The product when reacted with aqueous sodium hydroxide followed by dilute hydrochloric acid forms 2-Hydroxybenzoic acid or salicylic acid.
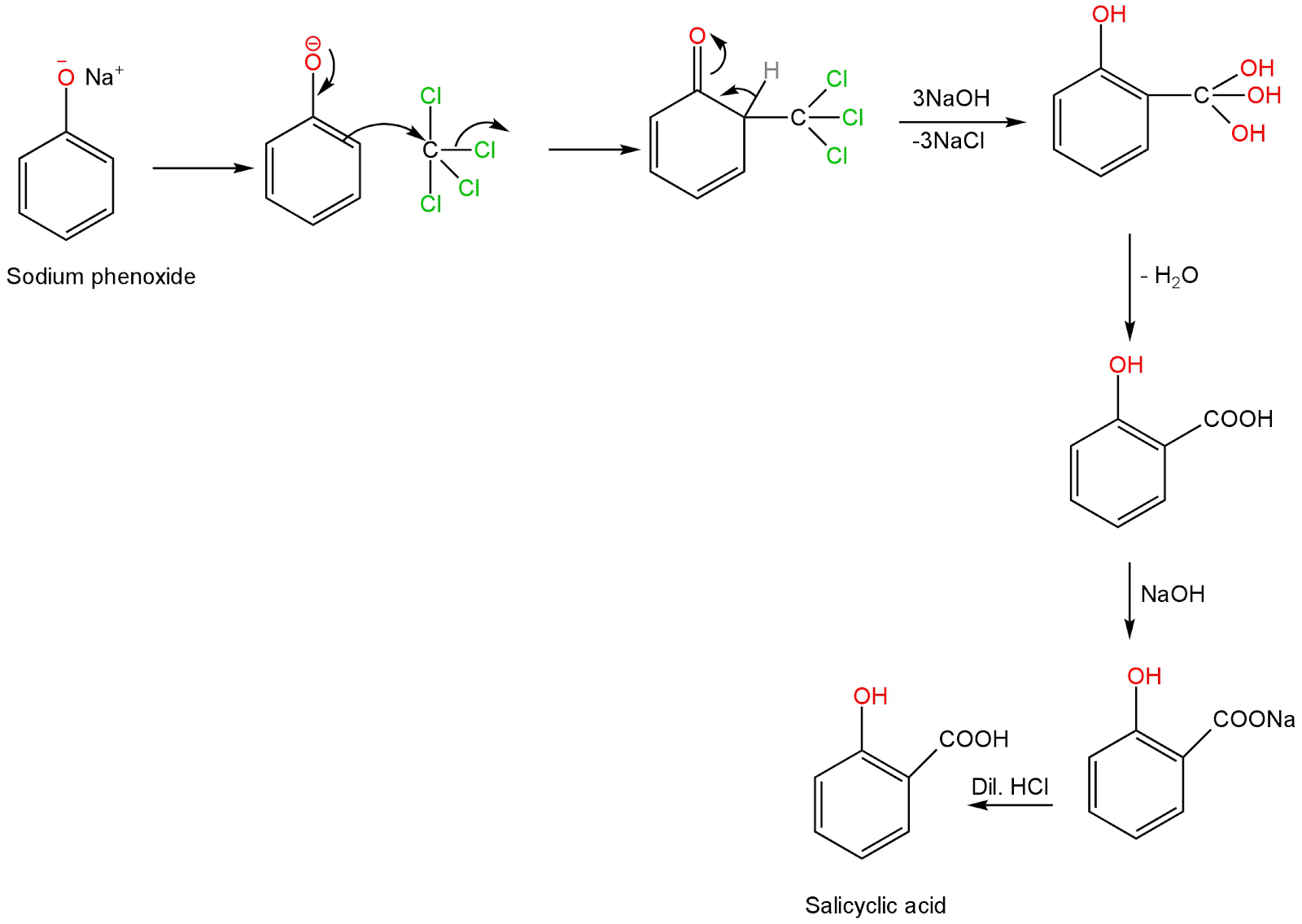
Image: Formation of salicylic acid from sodium phenoxide ion.
So, option A is correct.
Additional Information: Reaction of phenol with chloroform in aqueous sodium or potassium hydroxide at temperature 340 K then hydrolysis of the resulting product forms 2-hydroxybenzaldehyde or salicylaldehyde as the primary product.
Note: Reimer-Tiemann reaction is the production of salicylic acid and salicylaldehyde using phenol as the reactant.
For the formation of salicylic acid, carbon tetrachloride is used as a reagent and for salicylaldehyde, chloroform is used as a reagent.
It is called the Reimer-Tiemann reaction.
Complete step-by-step answer:
In the reaction of phenol with carbon tetrachloride in aqueous sodium or potassium hydroxide, salicylic acid is the product.
This reaction occurs at 340 K temperature.
The IUPAC name of Salicylic acid is 2-hydroxybenzoic acid.
The mechanism of the reaction is as follows:
It is an electrophilic substitution reaction.
The first step involves the removal of the proton from phenol in aqueous sodium hydroxide giving sodium phenoxide as the product.

Image: Sodium phenoxide ion formation
The next step involves the attack of carbon tetrachloride at the ortho position of the benzene ring.
This leads to the replacement of three chlorine atoms with three hydroxide ions.
The removal of a molecule of water happens in the following step.
The product when reacted with aqueous sodium hydroxide followed by dilute hydrochloric acid forms 2-Hydroxybenzoic acid or salicylic acid.

Image: Formation of salicylic acid from sodium phenoxide ion.
So, option A is correct.
Additional Information: Reaction of phenol with chloroform in aqueous sodium or potassium hydroxide at temperature 340 K then hydrolysis of the resulting product forms 2-hydroxybenzaldehyde or salicylaldehyde as the primary product.
Note: Reimer-Tiemann reaction is the production of salicylic acid and salicylaldehyde using phenol as the reactant.
For the formation of salicylic acid, carbon tetrachloride is used as a reagent and for salicylaldehyde, chloroform is used as a reagent.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




