Resonance energy per benzene ring decreases in the order
A . Naphthalene > benzene > Anthracene > Phenanthrene
B . Benzene > Naphthalene > Anthracene > Phenanthrene
C . Benzene > Naphthalene > Phenanthrene > Anthracene
D . All have equal resonance energy
Answer
262.8k+ views
Hint: For polycyclic aromatic hydrocarbons stability can be said to be proportional to resonance energy per benzene rings. So knowing the number of benzene rings each of the mentioned options are having, we can figure out the stability.
Complete step-by-step answer:
> The theoretical difference in molecular energy between resonance hybrid and the most stable resonance contributor is ( if the resonance contributor existed as a real molecule) is known as the resonance energy. We can also say that the stability gained by electron delocalization due to resonance versus the absence of such delocalization.
> Greater the resonance energy more will be the stability of the compound. This is because of the following two reasons:
- The contributing structures are all equivalent.
The number of contributing structures of roughly compatible energy is greater.
1) Here is the structure of Benzene :
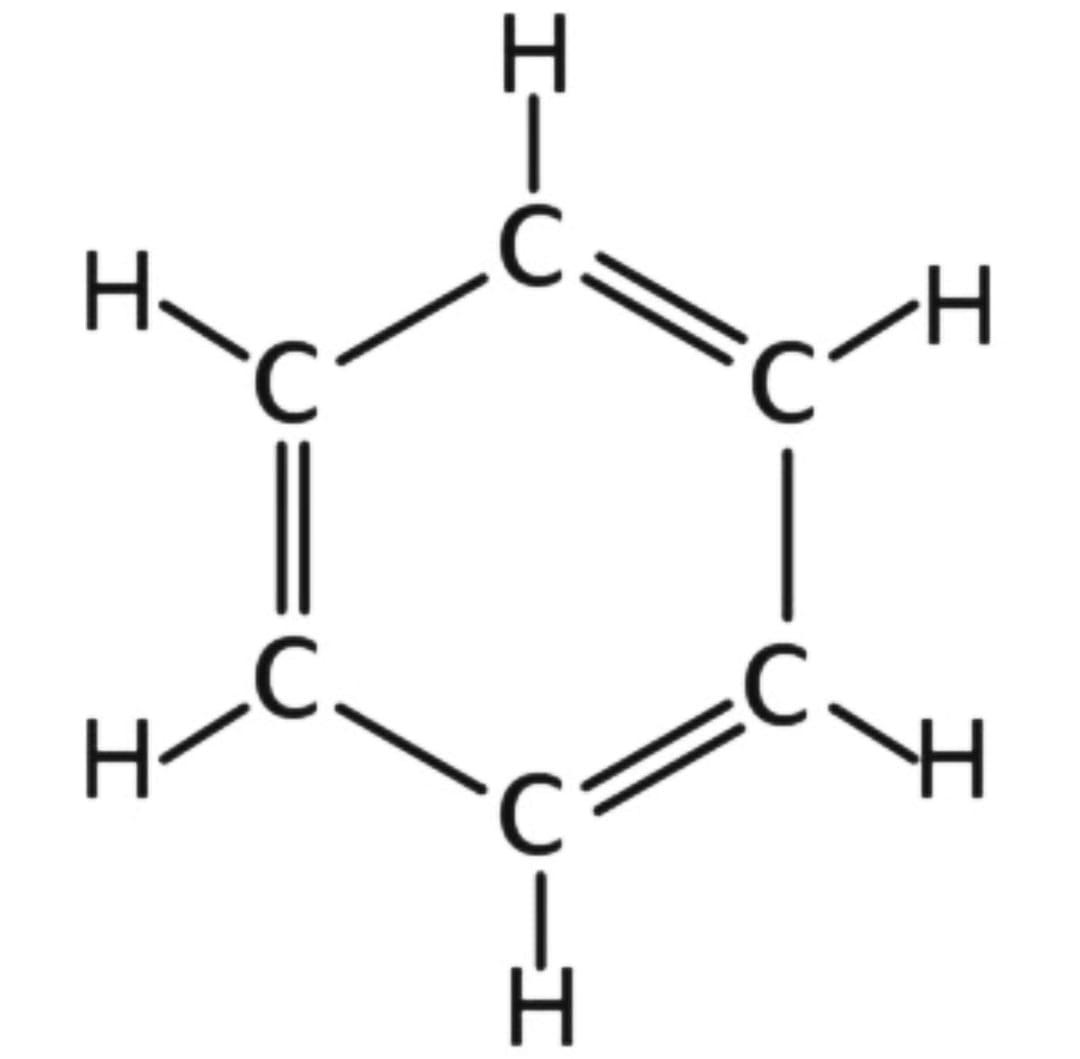
The resonance energy of benzene is 36 Kcal/mol, because there is one benzene ring.
2) Here is the structure of Naphthalene :
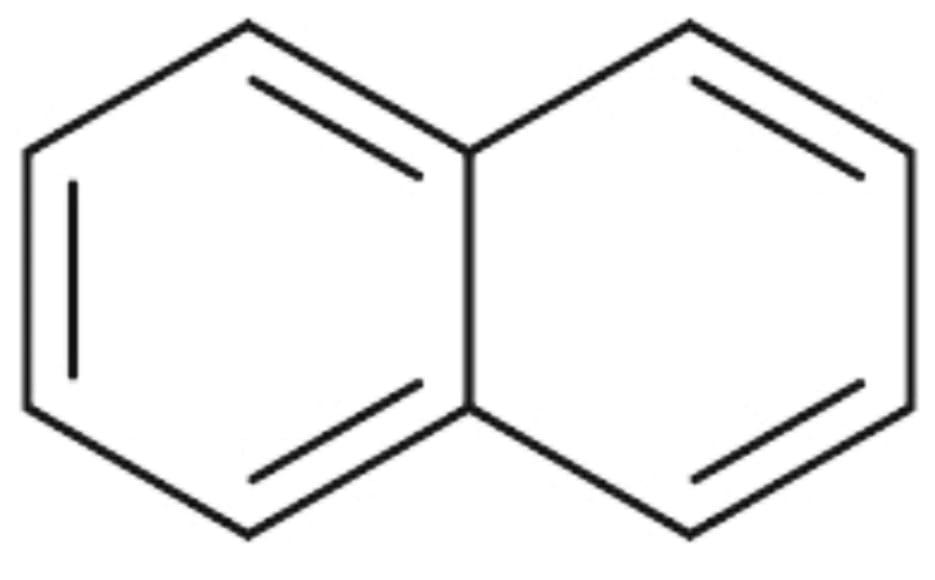
The resonance energy of Naphthalene is 30.5 Kcal/mol. There are two aromatic rings in Naphthalene.
3) The structure of Phenanthrene :
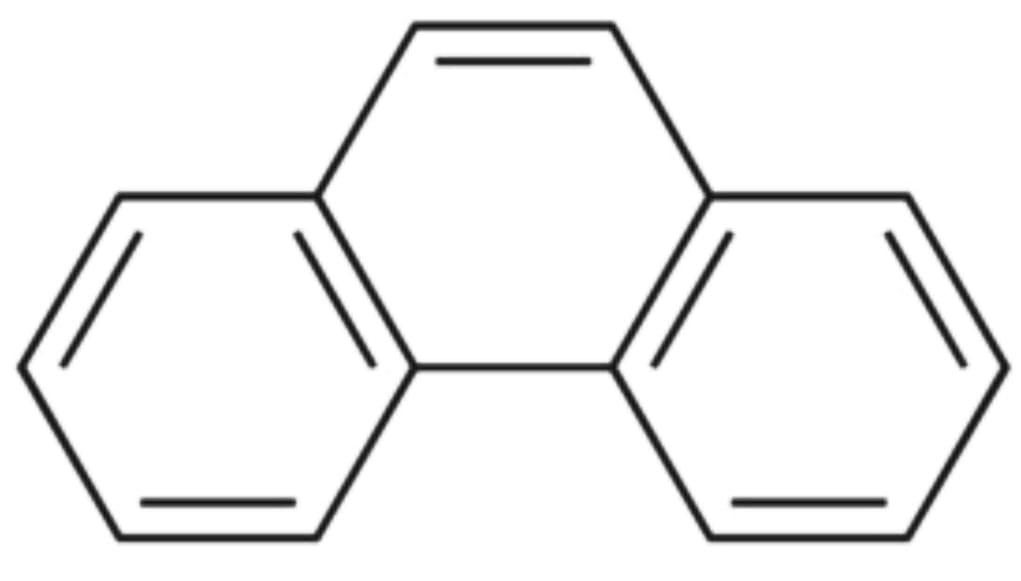
The resonance energy of Phenanthrene is 30.3 Kcal/mol. There are three aromatic rings in Phenanthrene.
4) The structure of Anthracene :
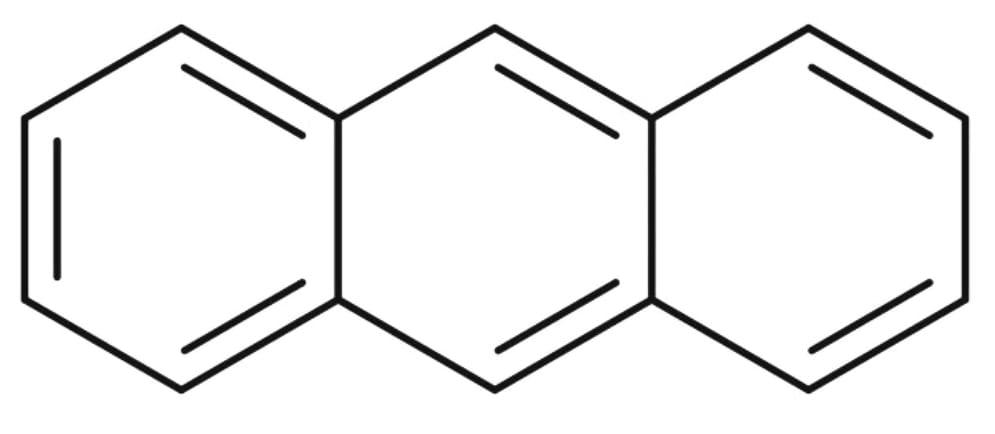
The resonance energy of Anthracene is 28 Kcal/mol. There are three aromatic rings in Anthracene.
Therefore the order of resonance energy as per the benzene ring in the decreasing order is : Benzene > Naphthalene > Phenanthrene > Anthracene. So the correct answer is Option C.
Additional Information:
The resonance energy of a compound is a measure of the extra stability of the conjugated system compared to the bonding number of isolated double bonds.
Note: We know that stability can be compared only for isomeric or related compounds. In case of unsaturated hydrocarbons, it is compared only when they give the same hydrogenated products.
Complete step-by-step answer:
> The theoretical difference in molecular energy between resonance hybrid and the most stable resonance contributor is ( if the resonance contributor existed as a real molecule) is known as the resonance energy. We can also say that the stability gained by electron delocalization due to resonance versus the absence of such delocalization.
> Greater the resonance energy more will be the stability of the compound. This is because of the following two reasons:
- The contributing structures are all equivalent.
The number of contributing structures of roughly compatible energy is greater.
1) Here is the structure of Benzene :

The resonance energy of benzene is 36 Kcal/mol, because there is one benzene ring.
2) Here is the structure of Naphthalene :

The resonance energy of Naphthalene is 30.5 Kcal/mol. There are two aromatic rings in Naphthalene.
3) The structure of Phenanthrene :

The resonance energy of Phenanthrene is 30.3 Kcal/mol. There are three aromatic rings in Phenanthrene.
4) The structure of Anthracene :

The resonance energy of Anthracene is 28 Kcal/mol. There are three aromatic rings in Anthracene.
Therefore the order of resonance energy as per the benzene ring in the decreasing order is : Benzene > Naphthalene > Phenanthrene > Anthracene. So the correct answer is Option C.
Additional Information:
The resonance energy of a compound is a measure of the extra stability of the conjugated system compared to the bonding number of isolated double bonds.
Note: We know that stability can be compared only for isomeric or related compounds. In case of unsaturated hydrocarbons, it is compared only when they give the same hydrogenated products.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




