Question:
The IUPAC name of chloretone is:
(A) Trichloroacetone
(B) Trichloronitromethane
(C) 1, 1, 1-trichloro-2-methyl-2-propanone
(D) 1, 1, 1-trichloro-2-methyl-2-propanol
Answer
266.4k+ views
Hint: Chloretone has a hydroxyl group and a halide (chlorine) as a functional group. We can use –ol suffix to describe hydroxyl functional groups and use Halo- prefix to describe presence of halogen in IUPAC nomenclature.
Step by step answer:
First of all we should know about chloretone. Chloretone is also called chlorobutanol, or chlorbutol.
- It is an alcohol based preservative. We use it as a preservative, sedative, hypnotic and weak local anesthetic similar in nature to chloral hydrate. It has antibacterial and antifungal properties. Now, we will draw the structure of chloretone.
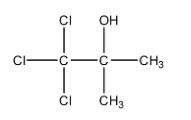
Let’s try to give the IUPAC name to this compound.
- To name this compound, we will first need to identify the longest carbon chain and we can see that there is a three carbon chain possible. We will start numbering the chain from the carbon that has a halogen group.
- There are 3 chlorine groups present at the first carbon chain and there is one alcohol group and one methyl group present on the second carbon of the chain. We will use –ol suffix to represent hydroxyl groups.
- So, we can say that the IUPAC name of chloretone is, 1, 1, 1- trichloro-2-methyl-2-propanol.
By this we can say that, option D is correct.
Note: Do not forget to arrange the substituent groups on the main carbon chain according to the alphabetical order in the process of naming a compound. Here, three chlorine atoms are present at the first carbon atom of the chain, so we will use trichloro- prefix instead of mentioning them separately.
Step by step answer:
First of all we should know about chloretone. Chloretone is also called chlorobutanol, or chlorbutol.
- It is an alcohol based preservative. We use it as a preservative, sedative, hypnotic and weak local anesthetic similar in nature to chloral hydrate. It has antibacterial and antifungal properties. Now, we will draw the structure of chloretone.

Let’s try to give the IUPAC name to this compound.
- To name this compound, we will first need to identify the longest carbon chain and we can see that there is a three carbon chain possible. We will start numbering the chain from the carbon that has a halogen group.
- There are 3 chlorine groups present at the first carbon chain and there is one alcohol group and one methyl group present on the second carbon of the chain. We will use –ol suffix to represent hydroxyl groups.
- So, we can say that the IUPAC name of chloretone is, 1, 1, 1- trichloro-2-methyl-2-propanol.
By this we can say that, option D is correct.
Note: Do not forget to arrange the substituent groups on the main carbon chain according to the alphabetical order in the process of naming a compound. Here, three chlorine atoms are present at the first carbon atom of the chain, so we will use trichloro- prefix instead of mentioning them separately.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




