Primary amines can be converted into alcohols by the action of
(A) Alkali
(B) Nitrous acid
(C) Reducing agent
(D) Oxidising agent
Answer
249.9k+ views
Hint: Primary amines are the amines in which the N of $N{{H}_{2}}$ group is attached to only one carbon. In the conversion of primary amines to alcohol, the amine $(-N{{H}_{2}})$ functional group is replaced by a hydroxyl ($-OH$) group.
Complete Step by Step Solution:
A primary amine can be converted into alcohol by the action of nitrous acid ($NaN{{O}_{2}}$ +$HCl$) on it. When nitrous acid ($NaN{{O}_{2}}$ +$HCl$) is treated with primary amines at a low temperature (273K – 278K), it gives rise to diazonium salt. When this diazonium salt is hydrolysed or treated with dilute acid, it forms alcohol. Along with the alcohol, side products like nitrogen (${{N}_{2}}$ ) gas and hydrochloric acid ($HCl$ ) are also formed.
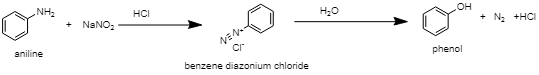
Correct Option: (B) Nitrous acid.
Additional Information: Alcohols have a wide range of applications in our daily lives. The paper we use for writing, the sugar we eat, and the sanitizers that we use to disinfect our hands are all made up of compounds that contain hydroxyl groups. The alcohols are soluble in water due to their ability to form hydrogen bonds in water. However, this solubility decreases with an increase in the size of alkyl or aryl groups.
Note: The well-known trihydric alcohol, glycerol ($C{{H}_{2}}(OH)-C{{H}_{2}}(OH)-C{{H}_{2}}(OH)$ )(IUPAC Name-Propane-1,2,3-triol) has various uses. It is used for the treatment of constipation. It can also be used as a solvent for flavours and food colours and for improving the performance of athletes.
Complete Step by Step Solution:
A primary amine can be converted into alcohol by the action of nitrous acid ($NaN{{O}_{2}}$ +$HCl$) on it. When nitrous acid ($NaN{{O}_{2}}$ +$HCl$) is treated with primary amines at a low temperature (273K – 278K), it gives rise to diazonium salt. When this diazonium salt is hydrolysed or treated with dilute acid, it forms alcohol. Along with the alcohol, side products like nitrogen (${{N}_{2}}$ ) gas and hydrochloric acid ($HCl$ ) are also formed.

Correct Option: (B) Nitrous acid.
Additional Information: Alcohols have a wide range of applications in our daily lives. The paper we use for writing, the sugar we eat, and the sanitizers that we use to disinfect our hands are all made up of compounds that contain hydroxyl groups. The alcohols are soluble in water due to their ability to form hydrogen bonds in water. However, this solubility decreases with an increase in the size of alkyl or aryl groups.
Note: The well-known trihydric alcohol, glycerol ($C{{H}_{2}}(OH)-C{{H}_{2}}(OH)-C{{H}_{2}}(OH)$ )(IUPAC Name-Propane-1,2,3-triol) has various uses. It is used for the treatment of constipation. It can also be used as a solvent for flavours and food colours and for improving the performance of athletes.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




