When primary amine is heated with CS2 in presence of excess mercuric chloride, it gives isothiocyanate. This reaction is called
A Hofmann bromide reaction
B Hofmann mustard oil reaction
C Carbylamine reaction
D Perkin reaction
Answer
249.3k+ views
Hint The substance that causes the black precipitate to form is almost completely insoluble in water. Cinnabar and metacinnabar are the two crystal forms of the dimorphic chemical. When it is used as a pigment, it is also known as vermillion.
Complete solution
In the Hofmann mustard oil reaction, isothiocyanates are created when primary amines are heated with alcoholic carbon disulfide and subsequently heated with excessive mercuric chloride. The reaction produces a strong odour that is brought on by these isothiocyanates. This odour is reminiscent of mustard oil.
A test to distinguish between primary, secondary, and tertiary amines is the Hofmann mustard oil reaction.
Now, let's examine how each amine responds to the Hoffman mustard oil test.
I) None of the reagents react with tertiary amines.
ii) Dithiocarbamic acids are created when secondary amines and carbon disulfide interact. However, it does not interact with the mercuric chloride in the following step.
When primary amines are treated with carbon disulfide and then heated with too much mercury chloride, isothiocyanates are produced that give off a strong odour akin to that of mustard oil.
We notice that a dark, insoluble precipitate forms during the process. Actually, during the process, HgS is produced as this insoluble precipitate.
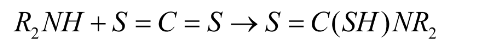
The correct option is (b) .
Note It should be noted that this test is employed to differentiate between primary, secondary, and tertiary amines. It's crucial to keep in mind that aromatic primary amines can pass this test as well. As a result, this test is unable to differentiate between aromatic and aliphatic amines.
Complete solution
In the Hofmann mustard oil reaction, isothiocyanates are created when primary amines are heated with alcoholic carbon disulfide and subsequently heated with excessive mercuric chloride. The reaction produces a strong odour that is brought on by these isothiocyanates. This odour is reminiscent of mustard oil.
A test to distinguish between primary, secondary, and tertiary amines is the Hofmann mustard oil reaction.
Now, let's examine how each amine responds to the Hoffman mustard oil test.
I) None of the reagents react with tertiary amines.
ii) Dithiocarbamic acids are created when secondary amines and carbon disulfide interact. However, it does not interact with the mercuric chloride in the following step.
When primary amines are treated with carbon disulfide and then heated with too much mercury chloride, isothiocyanates are produced that give off a strong odour akin to that of mustard oil.
We notice that a dark, insoluble precipitate forms during the process. Actually, during the process, HgS is produced as this insoluble precipitate.
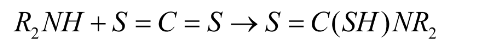
The correct option is (b) .
Note It should be noted that this test is employed to differentiate between primary, secondary, and tertiary amines. It's crucial to keep in mind that aromatic primary amines can pass this test as well. As a result, this test is unable to differentiate between aromatic and aliphatic amines.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




