p-Nitrobromobenzene can be converted to p-nitroaniline by using NaNH2. The reaction proceeds through the intermediate named
Answer
254.1k+ views
Hint: NaNH2 is an effective base. When its strong basicity doesn't have negative effects, it can occasionally be a great nucleophile. It is employed in elimination reactions as well as the deprotonation of weak acids. Alkynes, alcohols, and a variety of other functional groups with acidic protons, such as esters and ketones, will all be deprotonated by NaNH2.
Complete step by step solution:
Firstly, when NaNH2 reacts with p-Nitrobromobenzene, NaNH2 being a base will attack the most acidic hydrogen of p-Nitrobromobenzene. The carbon attached to Br will leave producing a carbanion.
In the next step, the bromine-carbon bond of p-Nitrobromobenzene will break and attach to the Na+ of NaNH2, leaving a positive charge on that carbon i.e, an empty orbital.
Now, two adjacent carbon having a positive and a negative charge creates instability and thus the carbon containing an electron pair will donate its electron pair to the adjacent carbon containing an empty orbital.
After this, a triple bond is formed between the two carbon atoms. The intermediate now formed is called benzyne. The nomenclature of the compound formed is 3,4-aryne.
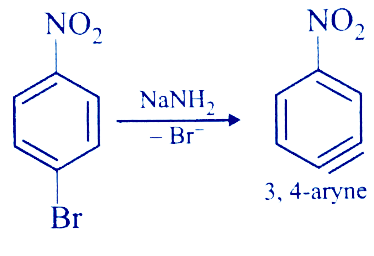
The correct answer is Benzyne.
Note: Because bases give up their electrons to form new covalent bonds, they are less likely to share them if they are more attracted to them. As a result we see that electronegativity is related to basicity. The greater the electronegativity of an atom, the less it is willing to share its electrons. Electronegativity is important to consider when comparing atoms in the same row of the periodic table.
Complete step by step solution:
Firstly, when NaNH2 reacts with p-Nitrobromobenzene, NaNH2 being a base will attack the most acidic hydrogen of p-Nitrobromobenzene. The carbon attached to Br will leave producing a carbanion.
In the next step, the bromine-carbon bond of p-Nitrobromobenzene will break and attach to the Na+ of NaNH2, leaving a positive charge on that carbon i.e, an empty orbital.
Now, two adjacent carbon having a positive and a negative charge creates instability and thus the carbon containing an electron pair will donate its electron pair to the adjacent carbon containing an empty orbital.
After this, a triple bond is formed between the two carbon atoms. The intermediate now formed is called benzyne. The nomenclature of the compound formed is 3,4-aryne.

The correct answer is Benzyne.
Note: Because bases give up their electrons to form new covalent bonds, they are less likely to share them if they are more attracted to them. As a result we see that electronegativity is related to basicity. The greater the electronegativity of an atom, the less it is willing to share its electrons. Electronegativity is important to consider when comparing atoms in the same row of the periodic table.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




