Phenolphthalein gives which of the following colours in basic salt?
(A) Blue
(B) Red
(C) Pink
(D) Purple
Answer
258.6k+ views
Hint: In the presence of acidic medium, phenolphthalein has a structure that does not impart any colour properties to the solution. The colour of phenolphthalein in basic medium is not a primary colour.
Step by step answer:
First of all, let’s get basic information about phenolphthalein. Phenolphthalein has a formula of ${{C}_{20}}{{H}_{14}}{{O}_{4}}$ . We should know that it is an organic compound of the phthalein family that is widely used as an acid-base indicator.
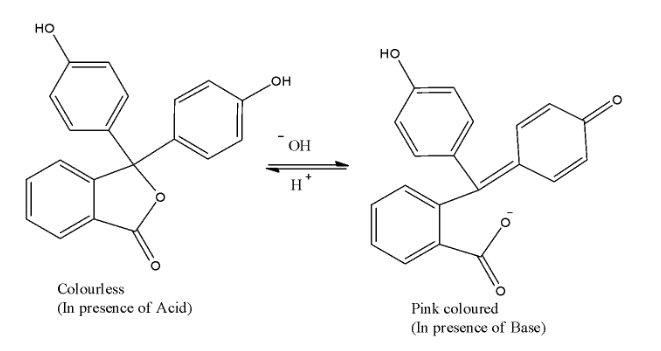
- The above structure is of phenolphthalein in both acidic medium and in basic medium.
- Phenolphthalein is primarily used as an indicator. It is used as an indicator in acid base titrations. When we add phenolphthalein in aqueous solution, it adopts at least four different states in aqueous solution as a result of pH changes.
- When we add phenolphthalein in acidic solution it exists in protonated form as shown in the reaction providing a colourless solution and when the solution turns basic in nature, the proton gets removed and a salt gets formed. This salt is a coloured one and we can see the solution turning pink due to this.
- So, we can say that phenolphthalein will give pink colour as a basic solution. And in the acidic solution, it will turn colourless.
- Thus, from the above discussion we can say that phenolphthalein will give pink colour in basic solution.
So, the correct option is C.
Note: Remember that in the basic medium, only one proton of phenolphthalein gets removed and results in a salt. So, two protons do not get removed in presence of basic medium even though there are similar two acidic protons are present in the phenolphthalein.
Step by step answer:
First of all, let’s get basic information about phenolphthalein. Phenolphthalein has a formula of ${{C}_{20}}{{H}_{14}}{{O}_{4}}$ . We should know that it is an organic compound of the phthalein family that is widely used as an acid-base indicator.

- The above structure is of phenolphthalein in both acidic medium and in basic medium.
- Phenolphthalein is primarily used as an indicator. It is used as an indicator in acid base titrations. When we add phenolphthalein in aqueous solution, it adopts at least four different states in aqueous solution as a result of pH changes.
- When we add phenolphthalein in acidic solution it exists in protonated form as shown in the reaction providing a colourless solution and when the solution turns basic in nature, the proton gets removed and a salt gets formed. This salt is a coloured one and we can see the solution turning pink due to this.
- So, we can say that phenolphthalein will give pink colour as a basic solution. And in the acidic solution, it will turn colourless.
- Thus, from the above discussion we can say that phenolphthalein will give pink colour in basic solution.
So, the correct option is C.
Note: Remember that in the basic medium, only one proton of phenolphthalein gets removed and results in a salt. So, two protons do not get removed in presence of basic medium even though there are similar two acidic protons are present in the phenolphthalein.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




