\[PC{l_5}\] reacts with propanone, to give
A. Vicinal dichloride
B. Propanal
C. propane chloride
D. Gem-dichloride
Answer
267k+ views
Hint: Phosphorus pentachloride is the name of the chemical compound PCl₅. It is a significant phosphorus chloride and is used as a chlorinating reagent. When this compound is treated with ketones, chlorination transpires at the carbonyl carbon.
Complete Step by Step Solution:
Phosphorus pentachloride is the chemical compound possessing the formula PCl5.
It is one of the considerably crucial phosphorus chlorides alongside\[PC{l_3}\] and\[POC{l_3}\].
\[PC{l_5}\] is utilised as a chlorinating reagent.
It has no colour and is sensitive to moisture and water.
Its structure is as follows:
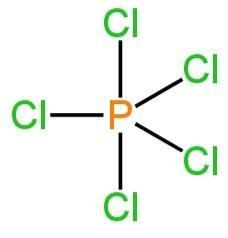
Image: Structure of phosphorus pentachloride
When it reacts with carbonyl groups like in ketones it leads to the conversion of C=O groups to \[CC{l_2}\]groups.
So, when it reacts with propanone it converts the C=O group to the CCl2 group.
Mechanism
1. The lone pair of oxygen in the carbonyl group attacks the phosphorus atom present in phosphorus pentachloride leading to the formation of a complex.
2. The complex formed when treated with a proton leads to the breaking of the carbon and oxygen bond. A carbocation is formed.
3. Carbocation is attacked by chloride ions present in the medium forming 2,2-dichloropropane and Phosphoryl chloride along with it which is a side product.
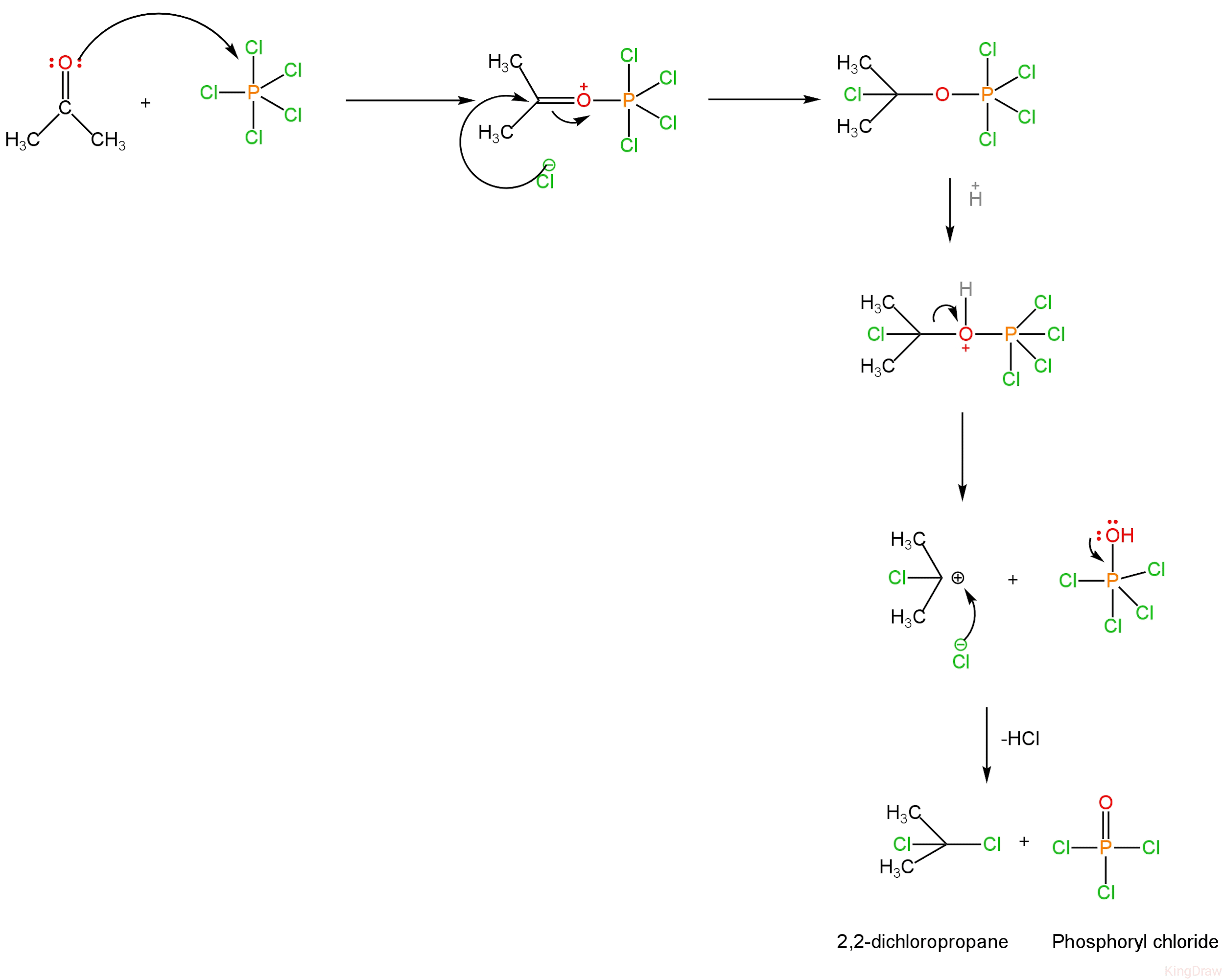
Image: Mechanism of chlorination of propanone.
As we know, 2,2-dichloropropane is a gem-dichloride. So, D is correct.
So, option D is correct.
Note: Phosphorus pentachloride also transforms alcohols into alkyl chlorides. This reaction is used in the preparation of alkyl chlorides. But thionyl chloride is more generally utilised in the laboratory as the resultant sulphur dioxide is a gas, and hence is more effortlessly isolated from the organic products than \[POC{l_3}\].
Complete Step by Step Solution:
Phosphorus pentachloride is the chemical compound possessing the formula PCl5.
It is one of the considerably crucial phosphorus chlorides alongside\[PC{l_3}\] and\[POC{l_3}\].
\[PC{l_5}\] is utilised as a chlorinating reagent.
It has no colour and is sensitive to moisture and water.
Its structure is as follows:

Image: Structure of phosphorus pentachloride
When it reacts with carbonyl groups like in ketones it leads to the conversion of C=O groups to \[CC{l_2}\]groups.
So, when it reacts with propanone it converts the C=O group to the CCl2 group.
Mechanism
1. The lone pair of oxygen in the carbonyl group attacks the phosphorus atom present in phosphorus pentachloride leading to the formation of a complex.
2. The complex formed when treated with a proton leads to the breaking of the carbon and oxygen bond. A carbocation is formed.
3. Carbocation is attacked by chloride ions present in the medium forming 2,2-dichloropropane and Phosphoryl chloride along with it which is a side product.

Image: Mechanism of chlorination of propanone.
As we know, 2,2-dichloropropane is a gem-dichloride. So, D is correct.
So, option D is correct.
Note: Phosphorus pentachloride also transforms alcohols into alkyl chlorides. This reaction is used in the preparation of alkyl chlorides. But thionyl chloride is more generally utilised in the laboratory as the resultant sulphur dioxide is a gas, and hence is more effortlessly isolated from the organic products than \[POC{l_3}\].
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




