Iodine oxidises sodium borohydride to give:
A) ${B_2}{H_6}$
B) Sodium hydride
C) $HI$
D) ${I_3}^{ - 1}$
Answer
260.1k+ views
Hint: Hydrogen borohydride is a minor reducing agent. It does selective reduction and it reduces substances by transfer of hydride ions (${H^ - }$).
Complete step by step solution:
-Sodium borohydride is an organic compound with molecular formula: $NaB{H_4}$. It is a salt consisting of a tetrahedral ($B{H_4}^ - $).
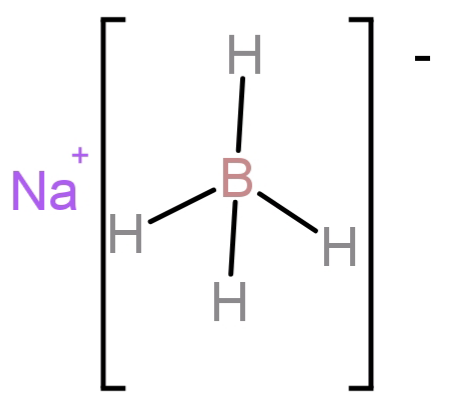
-It is an effective source of nucleophilic hydride ions in polar aprotic solvents like sulfolane, DMSO, DMF, HMPA or diglyme.
- So, it acts as a minor reducing agent and does selective reduction that is it reduces aldehydes and ketones to corresponding alcohols. It does not reduce double bonds. It reduces substance by transfer of the hydride ions.
-When one sodium borohydride molecule reacts with iodine, a borane ($B{H_3}$) molecule is produced along with sodium iodide ($NaI$) and hydrogen iodide ($HI$). Hydrogen iodide then reacts with another sodium borohydride molecule producing another borane and hydrogen iodide molecule. These 2 borane molecules thus do the bridging forming diborane (${B_2}{H_6}$). This is shown in the below reaction:
$NaB{H_4} + {I_2} \to B{H_3} + NaI + HI\xrightarrow{{NaB{H_4}}}B{H_3} + HI$
-This reaction takes place in the presence of Diglyme which acts as a catalyst.
-So, this reaction can also be written as:
$2NaB{H_4} + {I_2}\xrightarrow{{Diglyme}}2NaI + {B_2}{H_6} + {H_2}$
So, the correct option is: A) ${B_2}{H_6}$
Additional information: Diborane is found to be a rocket propellant because it is highly flammable. It undergoes a highly exothermic complete combustion. But it is unable to undergo complete combustion in a rocket engine and thus produces some amount of boron monoxide ($BO$).
Note: Before attempting read the question carefully about iodine oxidising sodium borohydride and not the other way round. And do not get confused between reducing agent (donates electron) and oxidising agent (accepts electron).
Complete step by step solution:
-Sodium borohydride is an organic compound with molecular formula: $NaB{H_4}$. It is a salt consisting of a tetrahedral ($B{H_4}^ - $).

-It is an effective source of nucleophilic hydride ions in polar aprotic solvents like sulfolane, DMSO, DMF, HMPA or diglyme.
- So, it acts as a minor reducing agent and does selective reduction that is it reduces aldehydes and ketones to corresponding alcohols. It does not reduce double bonds. It reduces substance by transfer of the hydride ions.
-When one sodium borohydride molecule reacts with iodine, a borane ($B{H_3}$) molecule is produced along with sodium iodide ($NaI$) and hydrogen iodide ($HI$). Hydrogen iodide then reacts with another sodium borohydride molecule producing another borane and hydrogen iodide molecule. These 2 borane molecules thus do the bridging forming diborane (${B_2}{H_6}$). This is shown in the below reaction:
$NaB{H_4} + {I_2} \to B{H_3} + NaI + HI\xrightarrow{{NaB{H_4}}}B{H_3} + HI$
-This reaction takes place in the presence of Diglyme which acts as a catalyst.
-So, this reaction can also be written as:
$2NaB{H_4} + {I_2}\xrightarrow{{Diglyme}}2NaI + {B_2}{H_6} + {H_2}$
So, the correct option is: A) ${B_2}{H_6}$
Additional information: Diborane is found to be a rocket propellant because it is highly flammable. It undergoes a highly exothermic complete combustion. But it is unable to undergo complete combustion in a rocket engine and thus produces some amount of boron monoxide ($BO$).
Note: Before attempting read the question carefully about iodine oxidising sodium borohydride and not the other way round. And do not get confused between reducing agent (donates electron) and oxidising agent (accepts electron).
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




