Why in the tetrahedral splitting, terms e and \[{t_2}\] are used, whereas, in octahedral splitting, terms as \[{e_g}\] and \[{t_{2g}}\] are used?
(A) Due to the approach of the ligands from the axis
(B) Due to the approach of the ligands in between the axis
(C) Due to the symmetry present in the octahedral system
(D) Due to the symmetry present in the tetrahedral system
Answer
255.3k+ views
Hint: If any system is symmetric or has centre of inversion symmetry, it is called gerade orbital, symbolised by g. while if no such symmetry exists, the g term is removed. Octahedral is a symmetric system while tetrahedral is not symmetric.
Complete step-by-step answer:
In an octahedral complex, six ligands are attached to the central transition metal. The d-orbital splits into two different levels. The bottom three energy levels are named as \[{d_{xy}}, {d_{yz}},{d_{xz}}\] and collectively referred to as \[{t_{2g}}\]. The two upper energy levels are named as \[{d_{{z^2}}}, {d_{{x^2} - {y^2}}}\], and collectively referred to as \[{e_g}\].
In a tetrahedral complex, four ligands are attached to the central metal. The d orbitals too split into two different energy levels. The top three consist of the \[{d_{xy}}, {d_{yz}}, {d_{xz}}\] orbitals and collectively referred as \[{t_2}\]. The bottom two consist of the \[{d_{{z^2}}}, {d_{{x^2} - {y^2}}}\] orbitals and are collectively referred to as e. The reason for this is due to poor orbital overlap between the metal and the ligand orbitals. The orbitals are directed on the axes, while the ligands are not as per the tetrahedral structure.
This difference in ‘g’ terms in both the structure is due to their symmetry. The word g stands for gerade which means symmetry, it is a German word. If the sign of the lobes remains the same, we call it a gerade orbital and if the signs are changed, the orbital is ungerade. In gerade, the centre of inversion symmetry is present.
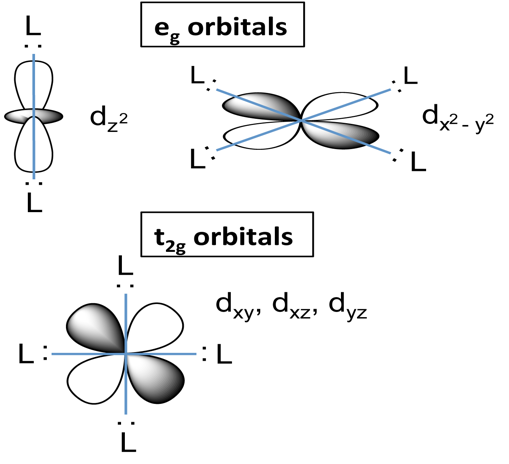
Tetrahedral complexes have no centre of symmetry and thus its orbital do not have g term in it. While in an octahedral system, g term is included because it is symmetric. Therefore, in the tetrahedral splitting, terms e and \[{t_2}\] are used, whereas, in octahedral splitting, terms as \[{e_g}\] and \[{t_{2g}}\] are used.
Hence, the correct option is (C).
Note: This can also be understood by Laporte selection rule which states that for a coordination complex that contains a centre of symmetry, only transitions which involve a change in parity of lobes are allowed. That is why octahedral is symmetric, hence allowed and vice versa for tetrahedral.
Complete step-by-step answer:
In an octahedral complex, six ligands are attached to the central transition metal. The d-orbital splits into two different levels. The bottom three energy levels are named as \[{d_{xy}}, {d_{yz}},{d_{xz}}\] and collectively referred to as \[{t_{2g}}\]. The two upper energy levels are named as \[{d_{{z^2}}}, {d_{{x^2} - {y^2}}}\], and collectively referred to as \[{e_g}\].
In a tetrahedral complex, four ligands are attached to the central metal. The d orbitals too split into two different energy levels. The top three consist of the \[{d_{xy}}, {d_{yz}}, {d_{xz}}\] orbitals and collectively referred as \[{t_2}\]. The bottom two consist of the \[{d_{{z^2}}}, {d_{{x^2} - {y^2}}}\] orbitals and are collectively referred to as e. The reason for this is due to poor orbital overlap between the metal and the ligand orbitals. The orbitals are directed on the axes, while the ligands are not as per the tetrahedral structure.
This difference in ‘g’ terms in both the structure is due to their symmetry. The word g stands for gerade which means symmetry, it is a German word. If the sign of the lobes remains the same, we call it a gerade orbital and if the signs are changed, the orbital is ungerade. In gerade, the centre of inversion symmetry is present.

Tetrahedral complexes have no centre of symmetry and thus its orbital do not have g term in it. While in an octahedral system, g term is included because it is symmetric. Therefore, in the tetrahedral splitting, terms e and \[{t_2}\] are used, whereas, in octahedral splitting, terms as \[{e_g}\] and \[{t_{2g}}\] are used.
Hence, the correct option is (C).
Note: This can also be understood by Laporte selection rule which states that for a coordination complex that contains a centre of symmetry, only transitions which involve a change in parity of lobes are allowed. That is why octahedral is symmetric, hence allowed and vice versa for tetrahedral.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




