In reversible isochoric change
A. $\Delta W = 0$
B. $\Delta Q = 0$
C. $\Delta T = 0$
D. $\Delta U = 0$
Answer
246.9k+ views
Hint: In this problem, to find out the correct option for a reversible isochoric change; we have to first find out the specific condition for the isochoric process and then analyze that condition with respect to different parameters such as work done, internal energy, temperature, etc., to give a correct result.
Complete answer:
Isochoric process in thermodynamics is a process during which the volume of a system remains constant that’s why it is also referred to as a constant-volume process.
A reversible process in thermodynamics is a process in which both the system and the surroundings return to their original state without any external effect on the universe.
According to the question, An Isochoric change to be reversible, the system must be maintained in thermal equilibrium and heat must be supplied in a quasi-static manner.
Also, we know that Pressure-Volume Work in thermodynamics is defined as: -
At constant pressure, $Workdone = W = P\Delta V$ … (1)
Graphically, an isochoric process can be represented as: -
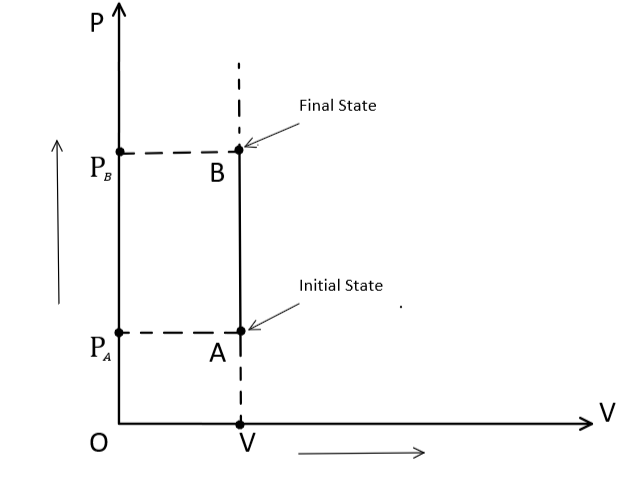
Also, By definition of an isochoric process, $\Delta V = 0$
So, from eq. (1), we get
$ \Rightarrow W = P\Delta V = 0$
Thus, In a reversible isochoric change $\Delta W = 0$. Hence, the correct option is (A) $\Delta W = 0$
Note: It is crucial to carefully consider all the given possibilities i.e., work done, internal energy, temperature, etc., in order to provide a comprehensive explanation because this is a theoretical conceptual problem. Always keep in mind to give specific justifications with the help of graphs in support of your explanation while writing an answer for this type of conceptual problem.
Complete answer:
Isochoric process in thermodynamics is a process during which the volume of a system remains constant that’s why it is also referred to as a constant-volume process.
A reversible process in thermodynamics is a process in which both the system and the surroundings return to their original state without any external effect on the universe.
According to the question, An Isochoric change to be reversible, the system must be maintained in thermal equilibrium and heat must be supplied in a quasi-static manner.
Also, we know that Pressure-Volume Work in thermodynamics is defined as: -
At constant pressure, $Workdone = W = P\Delta V$ … (1)
Graphically, an isochoric process can be represented as: -

Also, By definition of an isochoric process, $\Delta V = 0$
So, from eq. (1), we get
$ \Rightarrow W = P\Delta V = 0$
Thus, In a reversible isochoric change $\Delta W = 0$. Hence, the correct option is (A) $\Delta W = 0$
Note: It is crucial to carefully consider all the given possibilities i.e., work done, internal energy, temperature, etc., in order to provide a comprehensive explanation because this is a theoretical conceptual problem. Always keep in mind to give specific justifications with the help of graphs in support of your explanation while writing an answer for this type of conceptual problem.
Recently Updated Pages
JEE Mains Counselling 2026: Dates, Process, and Seat Allotment

JEE Mains Score Card 2026 Download – Check Marks and Rank

JEE Main 2026 Session 2 City Intimation Slip Out Now: Here's Your Direct Download Link

JEE Main 2026 Expected Cutoff: General, OBC, EWS, SC, ST Qualifying Marks & Score

JEE Main Sample Papers 2026 with Solutions - FREE PDF Download

Formulas Explained: Essential Math & Science Formulas

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

Understanding the Angle of Deviation in a Prism

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

CBSE Notes Class 11 Physics Chapter 4 - Laws of Motion - 2025-26




