The slopes of isothermal and adiabatic curves are related as:
A) Isothermal curve slope = Adiabatic curve slope
B) Isothermal curve slope = $\gamma \times $adiabatic slope
C) Adiabatic curve slope = $\gamma \times $isothermal slope
D) Adiabatic curve slope = $\dfrac{1}{2} \times $isothermal curve slope
Answer
567.4k+ views
Hint: Before we understand about these processes, it is important to understand the significance of the term slope. The slope represents the steepness of the curve, which is represented by the ratio of how high the curve is moving to the width of the curve. Hence, if we draw a tangent at the curve, the slope is given by tan of the angle made by the tangent with the horizontal.
The slope of the tangent drawn to a curve is equal to the derivative of the curve at the point where it touches the curve. Thus,
$Slope = \tan \theta = \dfrac{{dy}}{{dx}}$
Complete step by step answer:
Let us understand the meaning of the isothermal and adiabatic process in a thermodynamic system.
Isothermal process is a process that takes place in the system under constant temperature. The equation that represents an isothermal process is –
$PV = C$
where P = pressure and V = volume and C = constant
Adiabatic process is a process which takes place with zero heat transfer from the system or surroundings. The equation that represents an adiabatic process is –
$P{V^\gamma } = C$
where C = constant and $\gamma $ is the ratio of specific heats calculated at constant pressure and constant volume.
$\gamma = \dfrac{{{C_p}}}{{{C_v}}}$
Plotting these two processes on a pressure v/s volume graph, we obtain as follows:
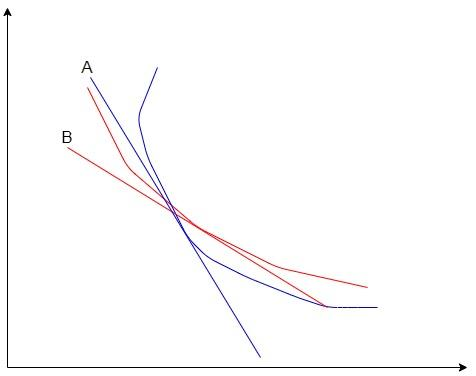
The red curve represents the isothermal process and the blue curve represents the adiabatic process. A and B are the slopes of the isothermal and adiabatic curves respectively. We can see from the graph, that the slope of adiabatic is higher than that of the isothermal process.Let us prove that.
If we differentiate the equation of the isothermal process, we obtain the slope of the line B in the above graph.
$PV = C$
Differentiating with respect to V,
$V\dfrac{{dP}}{{dV}} + P\dfrac{{dV}}{{dV}} = 0$
$ \Rightarrow V\dfrac{{dP}}{{dV}} = - P\dfrac{{dV}}{{dV}}$
$ \Rightarrow \dfrac{{dP}}{{dV}} = - \dfrac{P}{V}$
Hence, the slope of line A = $ - \dfrac{P}{V}$
Similarly, differentiating the equation for adiabatic process, we get the slope of line B.
$P{V^\gamma } = C$
Differentiating with respect to V,
$P\dfrac{{d\left( {{V^\gamma }} \right)}}{{dV}} + {V^\gamma }\dfrac{{dP}}{{dV}} = 0$
$ \Rightarrow {V^\gamma }\dfrac{{dP}}{{dV}} = - P\dfrac{{d\left( {{V^\gamma }} \right)}}{{dV}}$
$ \Rightarrow {V^\gamma }\dfrac{{dP}}{{dV}} = - P\gamma {V^{\gamma - 1}}$
\[\Rightarrow \dfrac{{dP}}{{dV}} = \dfrac{{ - P\gamma {V^{\gamma - 1}}}}{{{V^\gamma }}} = - P\gamma {V^{\gamma - 1 - \gamma }} = - P\gamma {V^{ - 1}}\]
\[\therefore \dfrac{{dP}}{{dV}} = - \gamma \dfrac{P}{V}\]
Comparing the slopes, we see that the slope of the adiabatic curve is $\gamma $ times the slope of the isothermal curve.
Hence, the correct option is Option C.
Note: The value $\gamma $ is referred to as the adiabatic index. If nothing is given, the value of adiabatic index for normal conditions of air should be taken as 1.4 approximately, while solving problems in adiabatic processes.
The slope of the tangent drawn to a curve is equal to the derivative of the curve at the point where it touches the curve. Thus,
$Slope = \tan \theta = \dfrac{{dy}}{{dx}}$
Complete step by step answer:
Let us understand the meaning of the isothermal and adiabatic process in a thermodynamic system.
Isothermal process is a process that takes place in the system under constant temperature. The equation that represents an isothermal process is –
$PV = C$
where P = pressure and V = volume and C = constant
Adiabatic process is a process which takes place with zero heat transfer from the system or surroundings. The equation that represents an adiabatic process is –
$P{V^\gamma } = C$
where C = constant and $\gamma $ is the ratio of specific heats calculated at constant pressure and constant volume.
$\gamma = \dfrac{{{C_p}}}{{{C_v}}}$
Plotting these two processes on a pressure v/s volume graph, we obtain as follows:

The red curve represents the isothermal process and the blue curve represents the adiabatic process. A and B are the slopes of the isothermal and adiabatic curves respectively. We can see from the graph, that the slope of adiabatic is higher than that of the isothermal process.Let us prove that.
If we differentiate the equation of the isothermal process, we obtain the slope of the line B in the above graph.
$PV = C$
Differentiating with respect to V,
$V\dfrac{{dP}}{{dV}} + P\dfrac{{dV}}{{dV}} = 0$
$ \Rightarrow V\dfrac{{dP}}{{dV}} = - P\dfrac{{dV}}{{dV}}$
$ \Rightarrow \dfrac{{dP}}{{dV}} = - \dfrac{P}{V}$
Hence, the slope of line A = $ - \dfrac{P}{V}$
Similarly, differentiating the equation for adiabatic process, we get the slope of line B.
$P{V^\gamma } = C$
Differentiating with respect to V,
$P\dfrac{{d\left( {{V^\gamma }} \right)}}{{dV}} + {V^\gamma }\dfrac{{dP}}{{dV}} = 0$
$ \Rightarrow {V^\gamma }\dfrac{{dP}}{{dV}} = - P\dfrac{{d\left( {{V^\gamma }} \right)}}{{dV}}$
$ \Rightarrow {V^\gamma }\dfrac{{dP}}{{dV}} = - P\gamma {V^{\gamma - 1}}$
\[\Rightarrow \dfrac{{dP}}{{dV}} = \dfrac{{ - P\gamma {V^{\gamma - 1}}}}{{{V^\gamma }}} = - P\gamma {V^{\gamma - 1 - \gamma }} = - P\gamma {V^{ - 1}}\]
\[\therefore \dfrac{{dP}}{{dV}} = - \gamma \dfrac{P}{V}\]
Comparing the slopes, we see that the slope of the adiabatic curve is $\gamma $ times the slope of the isothermal curve.
Hence, the correct option is Option C.
Note: The value $\gamma $ is referred to as the adiabatic index. If nothing is given, the value of adiabatic index for normal conditions of air should be taken as 1.4 approximately, while solving problems in adiabatic processes.
Recently Updated Pages
Circuit Switching vs Packet Switching: Key Differences Explained

Dimensions of Pressure in Physics: Formula, Derivation & SI Unit

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Understanding Atomic Structure for Beginners

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2026-27

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26




