In buna-S, the symbol ‘Bu’ stands for:
(A) 1 – butene
(B) n – butene
(C) 2 – butene
(D) Butadiene
Answer
260.4k+ views
Hint: Buna – S is a random copolymer which is formed by the emulsion polymerization of a mixture of butadiene and styrene in the ratio of \[1:3\]. This reaction takes place in the presence of a peroxide catalyst at \[5^\circ \] C and therefore the product is called cold rubber. The rubber obtained is also called Styrene butadiene rubber (SBR) or commonly known as Buna – S.
Complete Step-by-Step Solution:
Buna – S is a more commonly used name for a polymer named styrene butadiene rubber. The structure of Buna – S is given by:
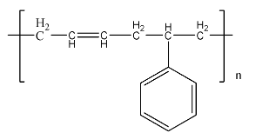
Buna – S is formed using the monomers butadiene and styrene. The reaction of this process can be given as:

In the word Buna – S, the Bu stands for Butadiene, since butadiene is one of the constituent monomers of the given polymer. Also, Na stands for sodium and S represents the monomer Styrene used for the synthesis of the given polymer.
Hence, Option D is the correct option.
Note: Buna -S can be derived from the monomers, butadiene and styrene using two different processes. These processes involve polymer formation from either a solution or an emulsion. The process solution is known as S-SBR while the process which prefers emulsion is known as E-SBR.
Complete Step-by-Step Solution:
Buna – S is a more commonly used name for a polymer named styrene butadiene rubber. The structure of Buna – S is given by:

Buna – S is formed using the monomers butadiene and styrene. The reaction of this process can be given as:

In the word Buna – S, the Bu stands for Butadiene, since butadiene is one of the constituent monomers of the given polymer. Also, Na stands for sodium and S represents the monomer Styrene used for the synthesis of the given polymer.
Hence, Option D is the correct option.
Note: Buna -S can be derived from the monomers, butadiene and styrene using two different processes. These processes involve polymer formation from either a solution or an emulsion. The process solution is known as S-SBR while the process which prefers emulsion is known as E-SBR.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




