IIdentify the product in following order
$3,4,5 - Tribromoaniline \xrightarrow[\left ( ii \right )H_3PO_4]{\left ( i \right )Diazotisation}$?
A. 3,4,5-Tribromobenzene
B. 1,2,3-Tribromobenzene
C. 2,4,6-Tribromobenzene
D. 3,4,5-Tribromo nitro benzene
E. 3,4,5-Tribromo phenol
Answer
266.7k+ views
Hint: 3,4,5-Tribromoaniline is a compound containing primary aromatic amine with three bromine groups at 3, 4 and 5 positions of aniline. Here in the first step diazotization is performed and then acid is given which will provide its proton in the reaction. Diazotization reaction is a type of coupling reaction for primary amine compounds.
Complete Step by Step Solution:
In the first step, a diazotisation reaction will take place where sodium nitrite and hydrochloric acid at \[{0^ \circ }\]C condition react to give nitrous acid, \[HONO\] which attacks the primary aromatic amine to give diazonium salt i.e., 3,4,5-Tribromobenzene diazonium salt. This then reacts with hypo phosphorus acid reagent which gives its hydrogen to the carbon of benzene ring where diazonium group is attached. So, from the final product nitrogen group is removed and hydrogen is present in its place. The name of the final compound is 1,2,3-tribromobenzene which is option B.
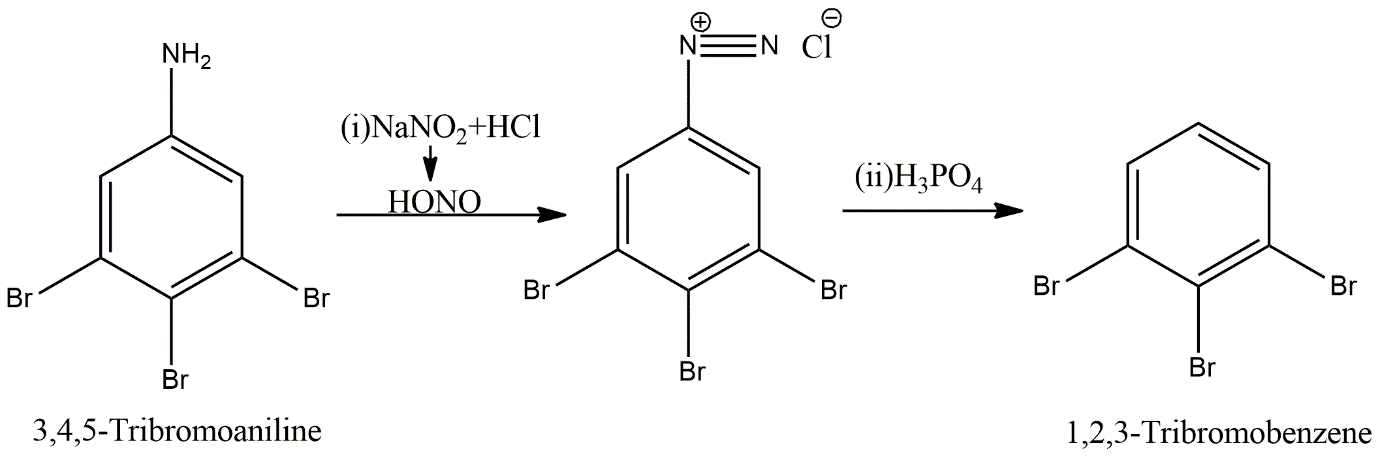
Image: Diazotisation reaction and further treatment with acid of 3,4,5-Tribromoaniline.
So, option B is correct. 2
Note: Ethanimine are the organic compounds having imine functional group i.e.,\[ - C = N - \]. This reacts with water and forms a compound containing a carbonyl functional group such as ketone or aldehyde. In this question acetonitrile is given so it forms acetaldehyde. Here, the overall number of carbons remains the same.
Complete Step by Step Solution:
In the first step, a diazotisation reaction will take place where sodium nitrite and hydrochloric acid at \[{0^ \circ }\]C condition react to give nitrous acid, \[HONO\] which attacks the primary aromatic amine to give diazonium salt i.e., 3,4,5-Tribromobenzene diazonium salt. This then reacts with hypo phosphorus acid reagent which gives its hydrogen to the carbon of benzene ring where diazonium group is attached. So, from the final product nitrogen group is removed and hydrogen is present in its place. The name of the final compound is 1,2,3-tribromobenzene which is option B.

Image: Diazotisation reaction and further treatment with acid of 3,4,5-Tribromoaniline.
So, option B is correct. 2
Note: Ethanimine are the organic compounds having imine functional group i.e.,\[ - C = N - \]. This reacts with water and forms a compound containing a carbonyl functional group such as ketone or aldehyde. In this question acetonitrile is given so it forms acetaldehyde. Here, the overall number of carbons remains the same.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




