If we use carbon tetrachloride (\[CC{l_4}\]) in the Riemer - Tiemann reaction in place of chloroform, the product formed is:
A. Salicylaldehyde
B. Phenolphthalein
C. Salicylic Acid
D. Cyclohexanol
Answer
266.7k+ views
Hint: The Riemer-Tiemann reaction involves the ortho-formylation of phenol. It takes place in the presence of chloroform and a strong hydroxide base. Under such conditions, phenol is converted into salicylaldehyde.
Complete Step by Step Solution:
The Riemer-Tiemann reaction is a chemical reaction in which phenol gets converted to salicylaldehyde. This occurs when phenol is reacted with chloroform (\[CHC{l_3}\]) in the presence of a strong base such as sodium hydroxide (\[NaOH\]) or potassium hydroxide (\[KOH\]).
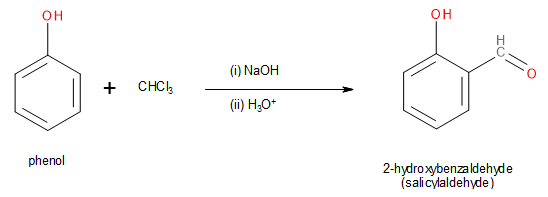
Image: Formation of Salicylaldehyde
However, the Riemer-Tiemann can be modified to produce phenolic acids such as salicylic acid by using carbon tetrachloride (\[CC{l_4}\]) instead of chloroform.
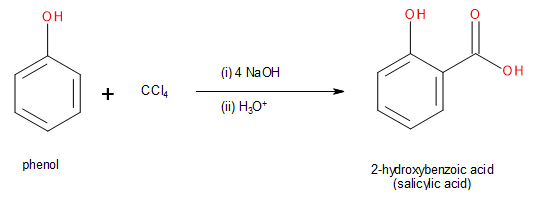
Image: Conversion of phenol into salicylic acid through Riemer-Tiemann reaction
The reaction proceeds in the following way.
First, the \[NaOH\] abstracts the acidic proton from phenol to give a phenoxide ion. The negative charge in the oxygen atom gets dispersed into the ring through the +R effect making the ring more nucleophilic.
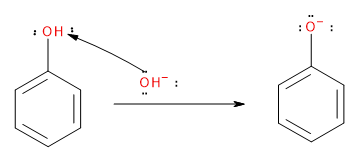
Image: Step 1: Abstraction of proton
The nucleophilic ring then attacks the electrophilic carbon atom of\[CC{l_4}\].
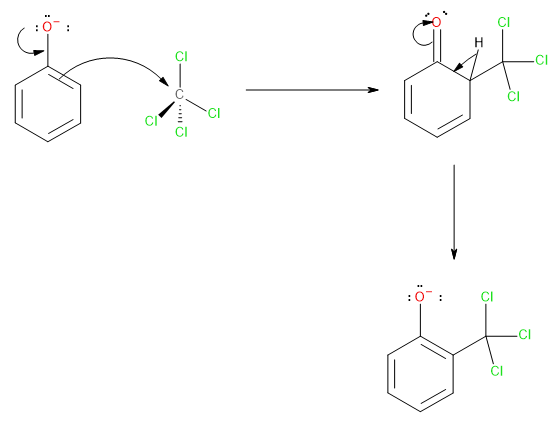
Image: Step 2
Each of the three chlorine atoms is removed by the sodium hydroxide leaving behind three hydroxyl groups attached to the carbon atom as shown here.
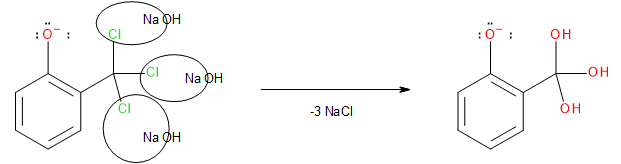
Image: Step 3
The product formed above undergoes spontaneous dehydration followed by acidic hydrolysis to give us the final product i.e., salicylic acid.
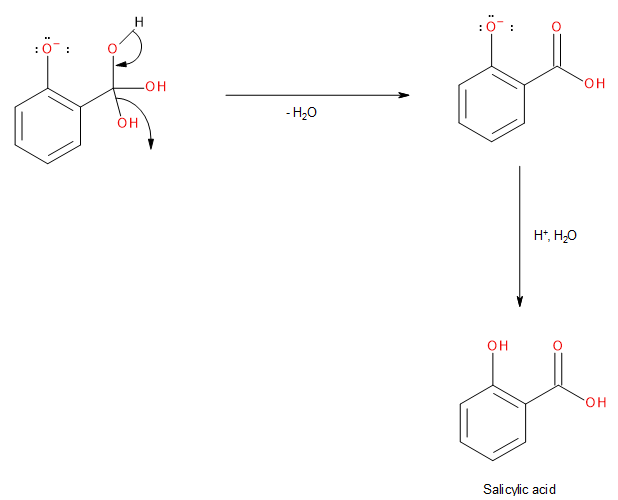
Image: Step 4
Thus, option C is the correct option.
Note: It must be remembered that the reaction mentioned above is only a variation of the Riemer-Tiemann reaction. The actual Riemer-Tiemann reaction leads to the formation of salicylaldehyde from phenol.
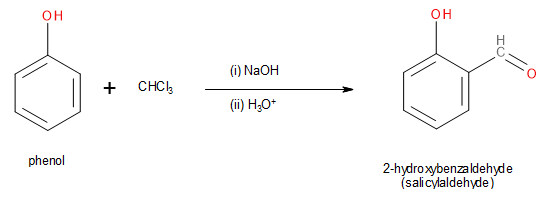
Image: Riemer-Tiemann reaction of phenol.
Complete Step by Step Solution:
The Riemer-Tiemann reaction is a chemical reaction in which phenol gets converted to salicylaldehyde. This occurs when phenol is reacted with chloroform (\[CHC{l_3}\]) in the presence of a strong base such as sodium hydroxide (\[NaOH\]) or potassium hydroxide (\[KOH\]).

Image: Formation of Salicylaldehyde
However, the Riemer-Tiemann can be modified to produce phenolic acids such as salicylic acid by using carbon tetrachloride (\[CC{l_4}\]) instead of chloroform.

Image: Conversion of phenol into salicylic acid through Riemer-Tiemann reaction
The reaction proceeds in the following way.
First, the \[NaOH\] abstracts the acidic proton from phenol to give a phenoxide ion. The negative charge in the oxygen atom gets dispersed into the ring through the +R effect making the ring more nucleophilic.

Image: Step 1: Abstraction of proton
The nucleophilic ring then attacks the electrophilic carbon atom of\[CC{l_4}\].

Image: Step 2
Each of the three chlorine atoms is removed by the sodium hydroxide leaving behind three hydroxyl groups attached to the carbon atom as shown here.

Image: Step 3
The product formed above undergoes spontaneous dehydration followed by acidic hydrolysis to give us the final product i.e., salicylic acid.

Image: Step 4
Thus, option C is the correct option.
Note: It must be remembered that the reaction mentioned above is only a variation of the Riemer-Tiemann reaction. The actual Riemer-Tiemann reaction leads to the formation of salicylaldehyde from phenol.

Image: Riemer-Tiemann reaction of phenol.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




