If true enter ${ 1 } $, else enter ${ 0 } $.
Covalent bonds are directional and ionic bonds are non-directional
(a) ${ 1 }$
(b) ${ 0 } $
(c) Both
(d) None of these
Answer
258.3k+ views
Hint: Covalent bond is defined as the bond which is formed by mutual sharing of electrons. Ionic bond is defined as the bond which is formed by the transfer of electrons from metal to non-metal. It occurs between oppositely charged ions.
Complete step-by-step solution:
> Covalent bonds are formed by the overlapping of atomic orbitals.So, the direction of overlapping gives direction of bonds,i.e, a shared pair of electron/ electrons are localized between two atoms. Hence, covalent bond is a directional bond.
> In ionic compounds, there is no overlapping of atomic orbitals. Each ion has its influence in all the directions. It means it is surrounded by a number of oppositely charged ions with no definite direction. Thus, ionic bonds are non-directional.
Figure:- Showing Lewis structure of covalent and ionic compounds
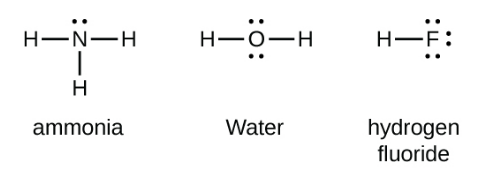
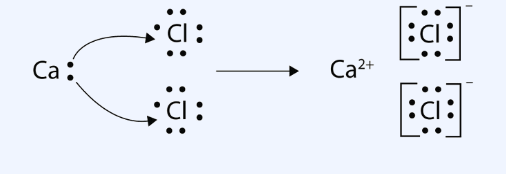
Hence, the above statement is true.Therefor answer is (A)
Additional Information: Polar Bonding:- A covalent bond formed between two different atoms, with different electronegativities is known as Polar covalent bond.
Non-polar bonding:- A covalent bond formed between two like atoms is known as Non-Polar bond. Here, the electronegativity difference is zero.
Note: The possibility for the mistake is that you can choose the option (b). You may think covalent bonds are non-directional and ionic bonds are directional. But it’s not true because in covalent bonds both sigma and pi bonding occurs and its directional while in ionic, an ion has the same attraction from all directions.
Complete step-by-step solution:
> Covalent bonds are formed by the overlapping of atomic orbitals.So, the direction of overlapping gives direction of bonds,i.e, a shared pair of electron/ electrons are localized between two atoms. Hence, covalent bond is a directional bond.
> In ionic compounds, there is no overlapping of atomic orbitals. Each ion has its influence in all the directions. It means it is surrounded by a number of oppositely charged ions with no definite direction. Thus, ionic bonds are non-directional.
Figure:- Showing Lewis structure of covalent and ionic compounds


Hence, the above statement is true.Therefor answer is (A)
Additional Information: Polar Bonding:- A covalent bond formed between two different atoms, with different electronegativities is known as Polar covalent bond.
Non-polar bonding:- A covalent bond formed between two like atoms is known as Non-Polar bond. Here, the electronegativity difference is zero.
Note: The possibility for the mistake is that you can choose the option (b). You may think covalent bonds are non-directional and ionic bonds are directional. But it’s not true because in covalent bonds both sigma and pi bonding occurs and its directional while in ionic, an ion has the same attraction from all directions.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




