Identify the pair in which the first structure is more stable than the second structure.
(A) 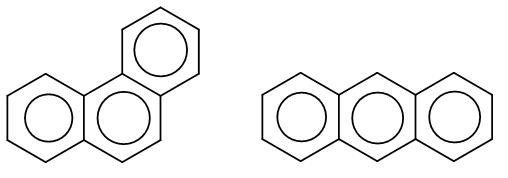
(B) 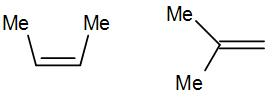
(C) 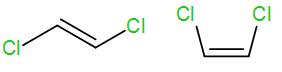
(D) 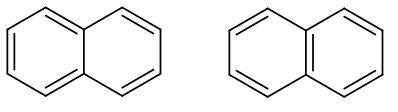
Answer
252.9k+ views
Hint: There are different factors that affect the stability of an organic compound. Here you have to consider the factors of resonance stabilization due to the formation of benzene like structures. Also, you have to consider the factors affecting the stability of an alkene.
Complete step by step solution:
To find the correct answer to this question, let us discuss the pairs in each of the options one by one.
Firstly, we have-
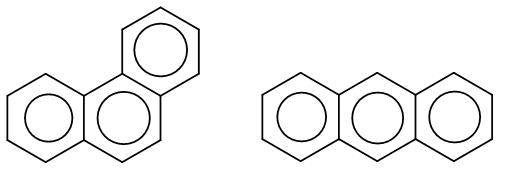
Here, the first structure is phenanthrene and the second structure is anthracene. Among these two there is an aromaticity difference which makes one of the more stable than the other.
In phenanthrene, the pi bonding is more efficient and it has two sextets whereas anthracene has just one sextet which makes phenanthrene more stable.
Then we have
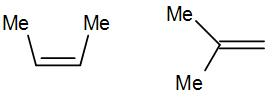
Here we have cis-alkene in the first structure and a geminal alkene i.e. the methyl groups are substituted to the same carbon.
The geminal structure is more stable than the cis or even trans alkene. The more substituted alkene is more stable according to Saytzeff’s rule a thus geminal structure is more stable than the cis structure.
Then we have
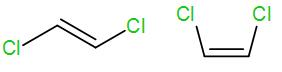
We know that trans alkene is more stable than cis alkenes. Here the first structure is a trans alkene and the second structure is a cis alkene.
The higher stability of trans alkene is due to lesser steric interaction among the substituted groups. In the cis structure, there is greater steric interaction between the two groups and thus causes de-stability.
And lastly, we have-
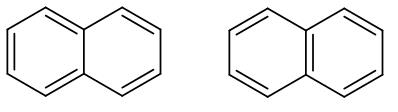
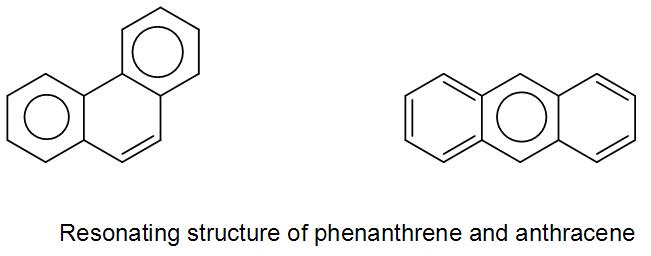
These are basically the two resonance structures of naphthalene. As we can see in both the structures the double bond is in conjugation. However, in the second structure as we can see there are two benzene rings therefore it has higher resonance energy compared to the first structure.
We can see from the above discussion that the correct answers are options (A) and (C).
Note: Saytzeff’s rule, also known as Zaitsev’s rule is a rule in organic chemistry which is used to find out the favoured alkene product in an elimination reaction. In a variety of elimination reactions, a general trend was observed in the resulting alkenes. Based on this general trend of the alkene products, Saytzeff’s rule was coined. We can write the actual statement of the rule as- "The alkene formed in the greatest amount is the one that corresponds to the removal of the hydrogen from the alpha-carbon having the fewest hydrogen substituents."
Complete step by step solution:
To find the correct answer to this question, let us discuss the pairs in each of the options one by one.
Firstly, we have-

Here, the first structure is phenanthrene and the second structure is anthracene. Among these two there is an aromaticity difference which makes one of the more stable than the other.
In phenanthrene, the pi bonding is more efficient and it has two sextets whereas anthracene has just one sextet which makes phenanthrene more stable.

Then we have
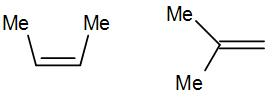
Here we have cis-alkene in the first structure and a geminal alkene i.e. the methyl groups are substituted to the same carbon.
The geminal structure is more stable than the cis or even trans alkene. The more substituted alkene is more stable according to Saytzeff’s rule a thus geminal structure is more stable than the cis structure.
Then we have

We know that trans alkene is more stable than cis alkenes. Here the first structure is a trans alkene and the second structure is a cis alkene.
The higher stability of trans alkene is due to lesser steric interaction among the substituted groups. In the cis structure, there is greater steric interaction between the two groups and thus causes de-stability.
And lastly, we have-

These are basically the two resonance structures of naphthalene. As we can see in both the structures the double bond is in conjugation. However, in the second structure as we can see there are two benzene rings therefore it has higher resonance energy compared to the first structure.
We can see from the above discussion that the correct answers are options (A) and (C).
Note: Saytzeff’s rule, also known as Zaitsev’s rule is a rule in organic chemistry which is used to find out the favoured alkene product in an elimination reaction. In a variety of elimination reactions, a general trend was observed in the resulting alkenes. Based on this general trend of the alkene products, Saytzeff’s rule was coined. We can write the actual statement of the rule as- "The alkene formed in the greatest amount is the one that corresponds to the removal of the hydrogen from the alpha-carbon having the fewest hydrogen substituents."
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




