Iodoform can be prepared from all except:
A. Acetaldehyde
B. \[3 - \]methyl\[ - 2 - \]butanone
C. Isobutyl alcohol
D. Acetophenone
Answer
259.8k+ views
Hint: Iodoform is an organoiodine compound with the formula \[{\text{CH}}{{\text{I}}_3}\].
We can synthesize iodoform using organic compounds having aldehyde or ketone group i.e carbonyl group having a methyl group on the side.
Complete step by step answer:
We can perform iodoform reaction using any of these four kinds of organic compounds \[i.e\]. a Methyl ketone (\[{\text{C}}{{\text{H}}_3}{\text{COR}}\]) or acetaldehyde (\[{\text{C}}{{\text{H}}_3}{\text{CHO}}\]) or ethanol (\[{\text{C}}{{\text{H}}_3}{\text{C}}{{\text{H}}_2}{\text{OH}}\]) or secondary alcohol. As all of the above mentioned organic compounds contain group or group.
Now, we will see the reaction scheme of iodoform reaction.
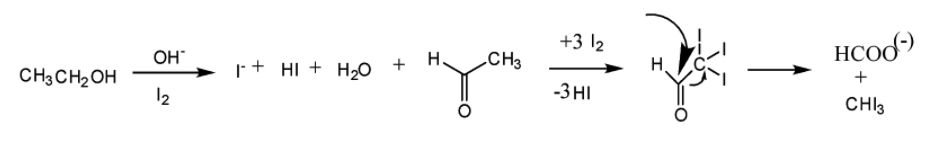
So, from the above discussion, we understand that the compound having an aldehydic group or -methyl ketone group will give this reaction.
Now, we will see the structure of the given compound.
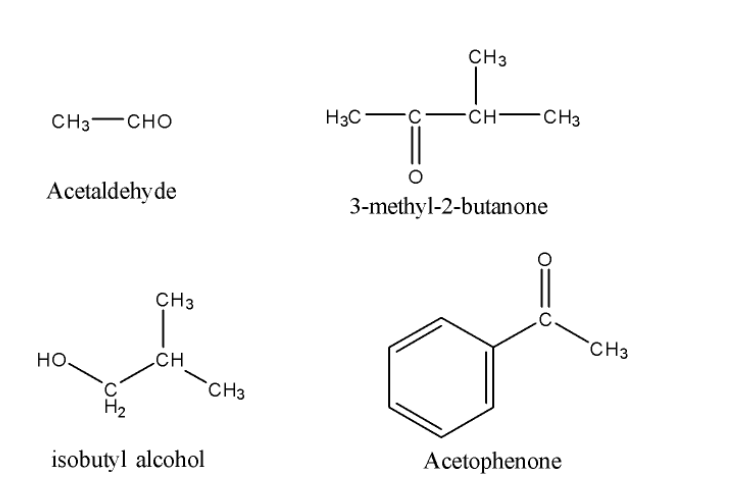
So, from the above structures, we can easily find out that all compounds except Isobutyl alcohol are having the required groups mentioned in the hint i.e aldehydic group or ketonic group with methyl on the side.
So, only the isobutyl alcohol will not give response to the iodoform test.
So, Iodoform can be prepared from all except isobutyl alcohol.
So, the correct option is option\[ - \left( {\mathbf{C}} \right).\]
Note:
1. Iodoform is a pale yellow colored, volatile substance and has a distinctive odor.
2. Like iodoform, there are chloroform and bromoform also. All the reactions go via the same mechanism.
3. While performing the iodoform reaction, care must be taken because of the corrosiveness of iodine.
We can synthesize iodoform using organic compounds having aldehyde or ketone group i.e carbonyl group having a methyl group on the side.
Complete step by step answer:
We can perform iodoform reaction using any of these four kinds of organic compounds \[i.e\]. a Methyl ketone (\[{\text{C}}{{\text{H}}_3}{\text{COR}}\]) or acetaldehyde (\[{\text{C}}{{\text{H}}_3}{\text{CHO}}\]) or ethanol (\[{\text{C}}{{\text{H}}_3}{\text{C}}{{\text{H}}_2}{\text{OH}}\]) or secondary alcohol. As all of the above mentioned organic compounds contain group or group.
Now, we will see the reaction scheme of iodoform reaction.

So, from the above discussion, we understand that the compound having an aldehydic group or -methyl ketone group will give this reaction.
Now, we will see the structure of the given compound.

So, from the above structures, we can easily find out that all compounds except Isobutyl alcohol are having the required groups mentioned in the hint i.e aldehydic group or ketonic group with methyl on the side.
So, only the isobutyl alcohol will not give response to the iodoform test.
So, Iodoform can be prepared from all except isobutyl alcohol.
So, the correct option is option\[ - \left( {\mathbf{C}} \right).\]
Note:
1. Iodoform is a pale yellow colored, volatile substance and has a distinctive odor.
2. Like iodoform, there are chloroform and bromoform also. All the reactions go via the same mechanism.
3. While performing the iodoform reaction, care must be taken because of the corrosiveness of iodine.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




