Hofmann's hypobromite reaction affords a method of:
- A. Preparing a tertiary amine
B. Preparing a mixture of amines
C. Stepping down a series
D. Stepping up a series
- A. Preparing a tertiary amine
Answer
263.1k+ views
Hint: Hoffmann’s hypobromite reaction mechanism is used and because of deprotonation reaction affords a mechanism of stepping down a series.
Complete step-by-step solution:Alkali is typically used as a strong base to attack the amide during the Hoffmann bromate reaction, which results in deprotonation and the subsequent formation of an anion. A primary amide can be changed into a primary amine with one less carbon atom using this process. This is done by heating the main amide with a solution of water, a strong base, and a halogen (chlorine or bromine).
Step 1: The strong base's hydroxide ion assaults the amide. Now that the amide has been deprotonated, water and the amide anion have been created.
Step 2: An alpha substitution reaction now takes place as the anion attacks the diatomic bromine. N-Bromamide and Br- anion are created as a result of the bromine-bromine bond breaking.
Step 3: The base now attacks the N-Bromamide once more, which causes its deprotonation and the production of water as well as the bromamide anion.
Step 4: This bromamide anion undergoes rearrangement so that the previously bonded ethyl group (or any other R- group) with the carbonyl carbon now binds with the nitrogen. The bromide anion that forms simultaneously departs from the complex. An isocyanate is produced as a result.
Step 5: A nucleophilic addition reaction causes carbamic acid to develop when water is added to the isocyanate.
step 6 : The carbamic acid now releases carbon dioxide, resulting in a negatively charged nitrogen linked to one hydrogen and the ethyl group in step 6. The necessary primary amine is created when this is protonated by the water.
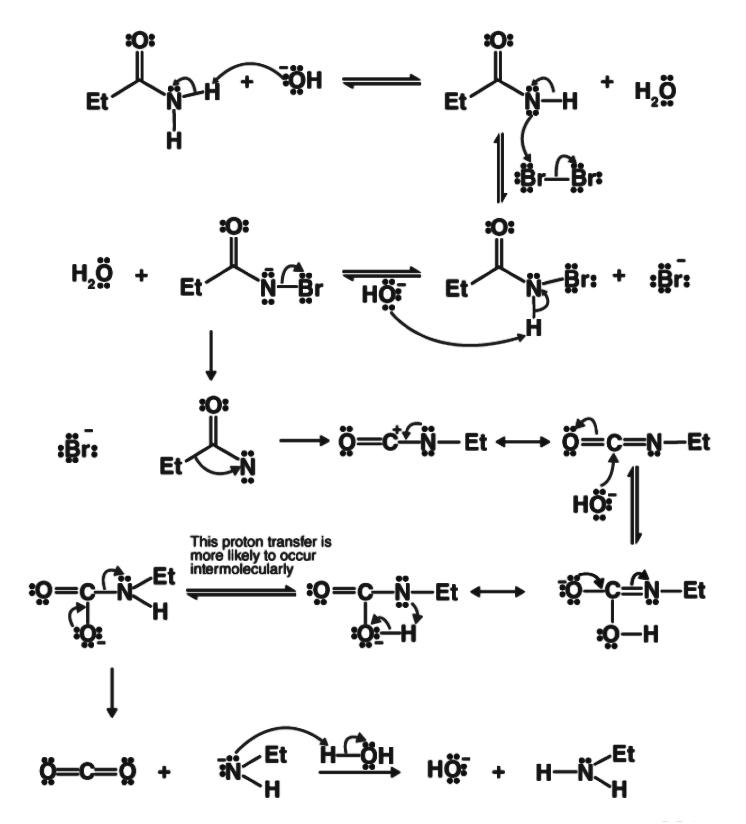
Hofmann's hypobromite reaction affords a method of stepping down a series.
\[C{{H}_{3}}CON{{H}_{2}}+B{{r}_{2}}+4KOH\to C{{H}_{3}}N{{H}_{2}}+{{K}_{2}}C{{O}_{3}}+2KBr+2{{H}_{2}}O\]
Hence, the correct option is option C which is stepping down a series.
Note: While writing the mechanism of the reaction we should always keep in mind the number of free radicals and the movement of the radicals.
Complete step-by-step solution:Alkali is typically used as a strong base to attack the amide during the Hoffmann bromate reaction, which results in deprotonation and the subsequent formation of an anion. A primary amide can be changed into a primary amine with one less carbon atom using this process. This is done by heating the main amide with a solution of water, a strong base, and a halogen (chlorine or bromine).
Step 1: The strong base's hydroxide ion assaults the amide. Now that the amide has been deprotonated, water and the amide anion have been created.
Step 2: An alpha substitution reaction now takes place as the anion attacks the diatomic bromine. N-Bromamide and Br- anion are created as a result of the bromine-bromine bond breaking.
Step 3: The base now attacks the N-Bromamide once more, which causes its deprotonation and the production of water as well as the bromamide anion.
Step 4: This bromamide anion undergoes rearrangement so that the previously bonded ethyl group (or any other R- group) with the carbonyl carbon now binds with the nitrogen. The bromide anion that forms simultaneously departs from the complex. An isocyanate is produced as a result.
Step 5: A nucleophilic addition reaction causes carbamic acid to develop when water is added to the isocyanate.
step 6 : The carbamic acid now releases carbon dioxide, resulting in a negatively charged nitrogen linked to one hydrogen and the ethyl group in step 6. The necessary primary amine is created when this is protonated by the water.

Hofmann's hypobromite reaction affords a method of stepping down a series.
\[C{{H}_{3}}CON{{H}_{2}}+B{{r}_{2}}+4KOH\to C{{H}_{3}}N{{H}_{2}}+{{K}_{2}}C{{O}_{3}}+2KBr+2{{H}_{2}}O\]
Hence, the correct option is option C which is stepping down a series.
Note: While writing the mechanism of the reaction we should always keep in mind the number of free radicals and the movement of the radicals.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




