Highest ionization potential in a period is shown by:
(A) Alkali metals
(B) Transition elements
(C) Halogens
(D) Noble gases
Answer
268.2k+ views
Hint: The closer and more tightly bound an electron is to the nucleus, the more difficult it will be to remove and higher will be its ionization potential. The ionization potential or ionization energy decreases from top to bottom in groups and increases from left to right across a period.
Complete step by step solution:
The ionization energy or ionization energy of an atom is the amount of energy that is required to remove an electron from a mole of atoms in the gas phase. In a periodic table, the ionization potential decreases from top to bottom in groups while it increases when crossing from right to left in a period. It can be understood by the image shown below,
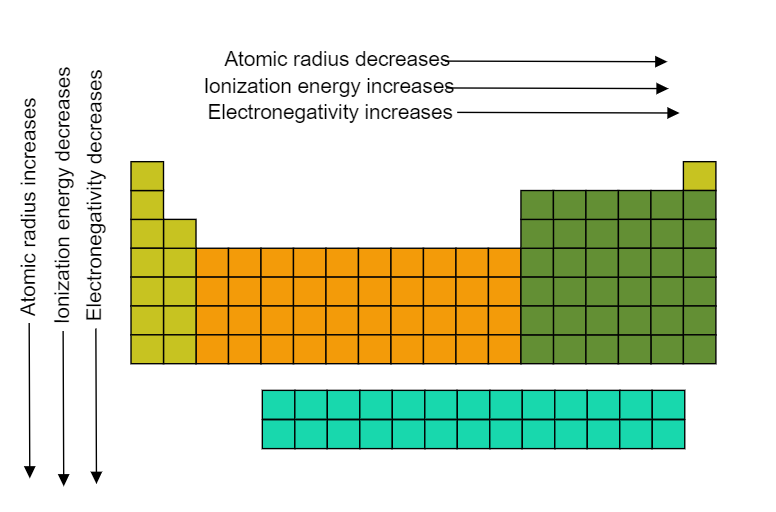
So, the groups further to the right of the periodic table would have greater ionization potential because they are more stable and they do not want to donate its electrons to other atoms and also there is the greater number of protons (positively charged) that attracts the electrons (negatively charged) which are present in the outer shell thereby requiring more energy to remove an electron from the shell.
Thus, concluding noble gases to be having the highest ionization potential. This is shown by the noble gases because an electron is to be removed from a completely filled orbital which further breaks the stable electronic configuration ns2np6 requiring a large amount of energy.
Hence, the correct option will be (D).
Note: Technically the noble gases have the largest ionization potential, but since they are special and it is not often seen that electrons would even be removed from these elements as they rarely react with other elements, you would usually get confused with halogens having the largest ionization potential as they are present before the noble gases in a period in the periodic table.
Complete step by step solution:
The ionization energy or ionization energy of an atom is the amount of energy that is required to remove an electron from a mole of atoms in the gas phase. In a periodic table, the ionization potential decreases from top to bottom in groups while it increases when crossing from right to left in a period. It can be understood by the image shown below,

So, the groups further to the right of the periodic table would have greater ionization potential because they are more stable and they do not want to donate its electrons to other atoms and also there is the greater number of protons (positively charged) that attracts the electrons (negatively charged) which are present in the outer shell thereby requiring more energy to remove an electron from the shell.
Thus, concluding noble gases to be having the highest ionization potential. This is shown by the noble gases because an electron is to be removed from a completely filled orbital which further breaks the stable electronic configuration ns2np6 requiring a large amount of energy.
Hence, the correct option will be (D).
Note: Technically the noble gases have the largest ionization potential, but since they are special and it is not often seen that electrons would even be removed from these elements as they rarely react with other elements, you would usually get confused with halogens having the largest ionization potential as they are present before the noble gases in a period in the periodic table.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
Understanding Electromagnetic Waves and Their Importance

JEE Main Colleges 2026: Complete List of Participating Institutes

Understanding the Angle of Deviation in a Prism

Formal charge on nitrogen and oxygen in NO3 ion are class 11 chemistry JEE_Main

Understanding Displacement and Velocity Time Graphs

Understanding Essential Formulas for Students

Other Pages
JEE Advanced 2026 Revision Notes for Vectors

NCERT Solutions For Class 11 Chemistry Chapter 7 Redox Reaction - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 5 Thermodynamics - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 9 Hydrocarbons - 2025-26

Electron Gain Enthalpy and Electron Affinity Explained

Degree of Dissociation: Meaning, Formula, Calculation & Uses




