For which of the following molecules is significant $\mu \ne 0$?
(A)  (B)
(B)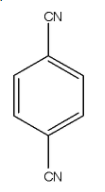 (C)
(C) 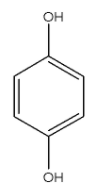 (D)
(D)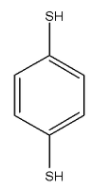
(A) Only (C)
(B) (C) and (D)
(C) Only (A)
(D) (A) and (B)
Answer
260.1k+ views
Hint: The dipole moment can be defined as a negative or positive charge multiplied to the distance between the centres of positive and negative charges. The direction of dipole moment is always toward the more electronegative atom. The dipole moment cancels out when the direction of the dipole moment is in the opposite direction.
Complete step by step solution:
The dipole moment can be defined as a negative or positive charge multiplied to the distance between the centres of positive and negative charges. It is denoted by$\mu $. It is denoted by the arrow with its tail at the positive centre and head pointing towards the negative end:
$(+\mapsto -)$
The dipole moment is directed towards the more electronegative atom.
Let us see all the options:
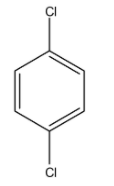
This compound is 1, 4-Dichlorobenzene. So, the chlorine is more electronegative atom than the carbon atom so the dipole moment will be towards the chlorine atom. So, both chlorines are in the opposite direction and the dipole moment will be cancelled out. Hence, it will have $\mu $ will be 0.
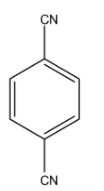
This compound is 1, 4-Dicyanobenzene. So, the carbon atom in the cyano group is more electronegative than the carbon atom of the benzene ring so the dipole moment will be towards the Cyano group. So, both Cyano is in the opposite direction and the dipole moment will be cancelled out. Hence, it will have $\mu $ will be 0.
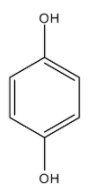
This compound is Benzene-1,4-diol. There are two conformers of this compound.

In these conformers both the groups are not in the opposite direction and the dipole moment will be towards the oxygen atom but it will not cancel out each other. Hence they will have $\mu \ne 0$.
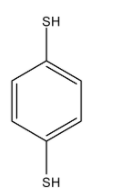
This compound is 1,4-Benzenedithiol. There are two conformers of this compound.
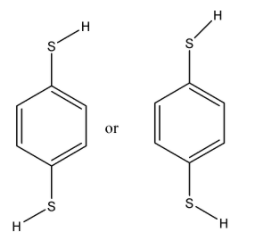
In these conformers both the groups are not in the opposite direction and the dipole moment will be towards the carbon atom but it will not cancel out each other. Hence they will have $\mu \ne 0$.
Therefore, the correct answer is an option (B)- (C) and (D).
Note: The determination of the dipole moment helps find the polarity of the molecule. The higher the dipole moment of the molecule, the higher is the polarity of the molecule. It can also be used in calculating the ionic character.
Complete step by step solution:
The dipole moment can be defined as a negative or positive charge multiplied to the distance between the centres of positive and negative charges. It is denoted by$\mu $. It is denoted by the arrow with its tail at the positive centre and head pointing towards the negative end:
$(+\mapsto -)$
The dipole moment is directed towards the more electronegative atom.
Let us see all the options:

This compound is 1, 4-Dichlorobenzene. So, the chlorine is more electronegative atom than the carbon atom so the dipole moment will be towards the chlorine atom. So, both chlorines are in the opposite direction and the dipole moment will be cancelled out. Hence, it will have $\mu $ will be 0.

This compound is 1, 4-Dicyanobenzene. So, the carbon atom in the cyano group is more electronegative than the carbon atom of the benzene ring so the dipole moment will be towards the Cyano group. So, both Cyano is in the opposite direction and the dipole moment will be cancelled out. Hence, it will have $\mu $ will be 0.

This compound is Benzene-1,4-diol. There are two conformers of this compound.

In these conformers both the groups are not in the opposite direction and the dipole moment will be towards the oxygen atom but it will not cancel out each other. Hence they will have $\mu \ne 0$.

This compound is 1,4-Benzenedithiol. There are two conformers of this compound.

In these conformers both the groups are not in the opposite direction and the dipole moment will be towards the carbon atom but it will not cancel out each other. Hence they will have $\mu \ne 0$.
Therefore, the correct answer is an option (B)- (C) and (D).
Note: The determination of the dipole moment helps find the polarity of the molecule. The higher the dipole moment of the molecule, the higher is the polarity of the molecule. It can also be used in calculating the ionic character.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




