For the given reaction \[(B)\xleftarrow{NaB{{H}_{4}}}PhCH=CH-CHO\xrightarrow[2){{H}_{3}}{{O}^{+}}]{1)LAH,ether}(A)\]
The products (A) and (B) are:
(A) 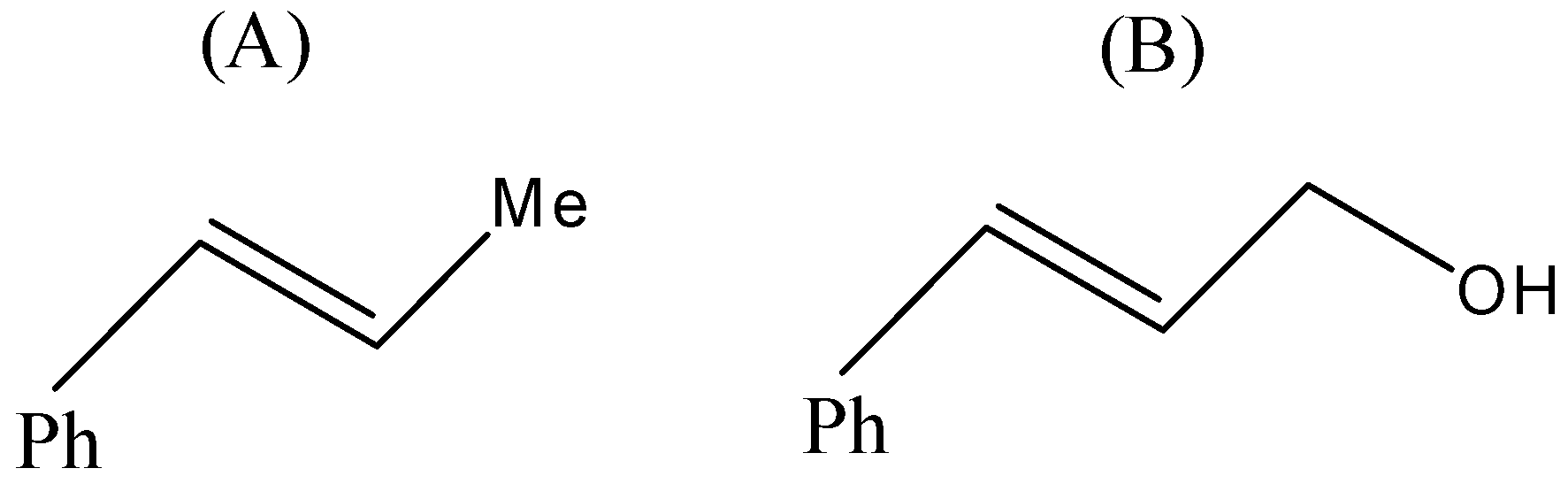
(B) 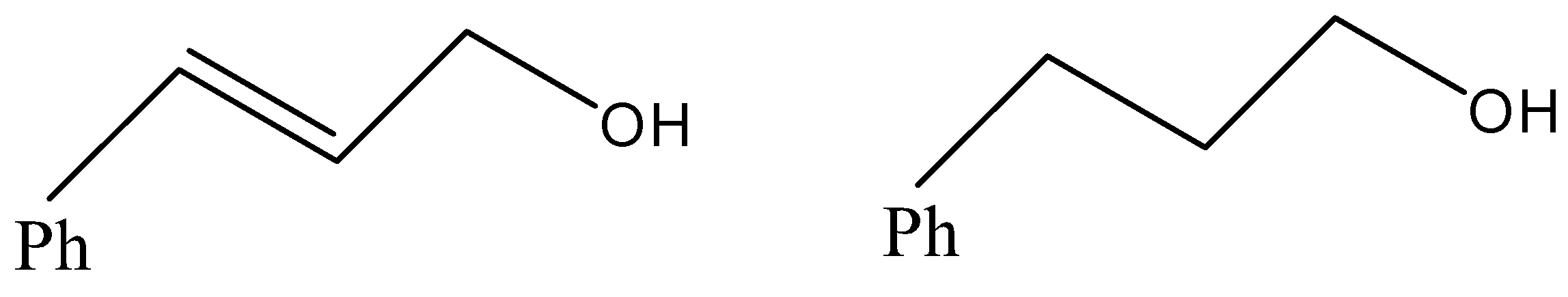
(C) 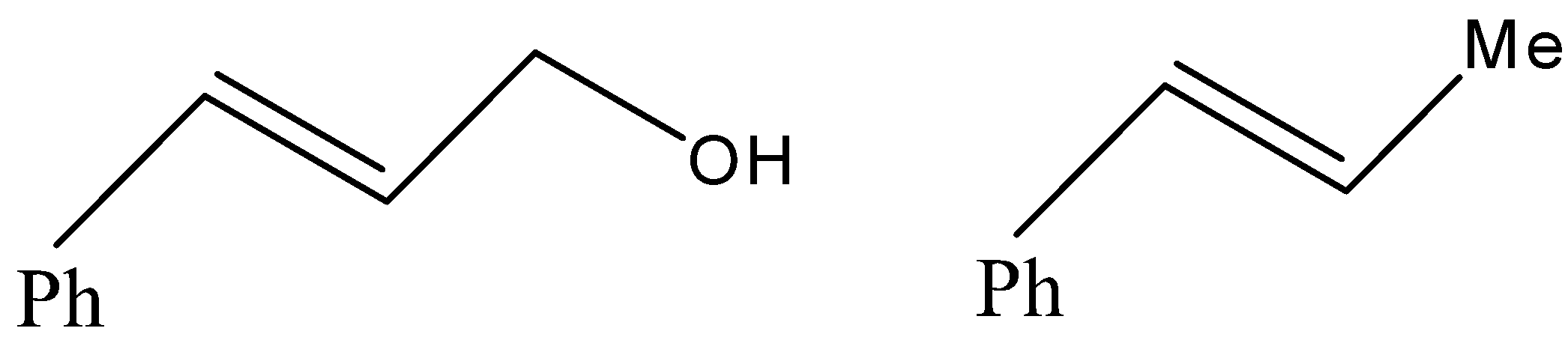
(D) 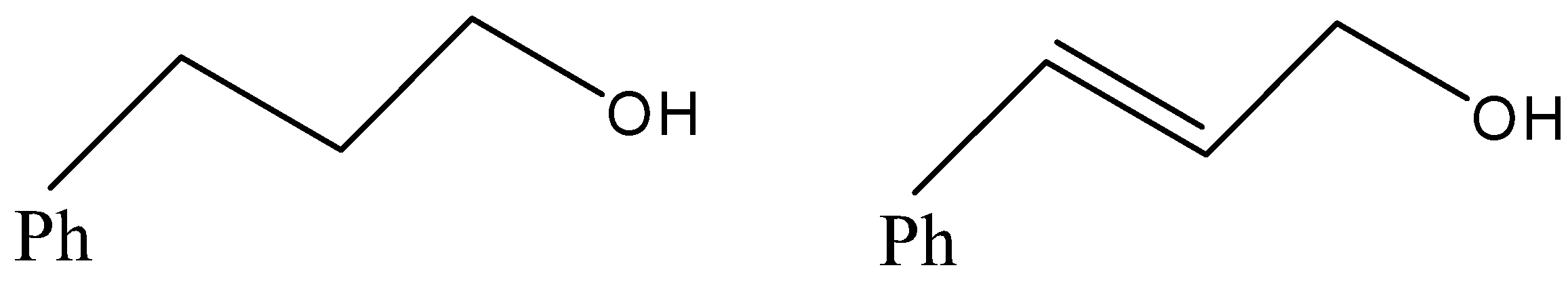
Answer
257.7k+ views
Hint: LAH stands for Lithium Aluminium Hydride and $NaB{{H}_{4}}$ is sodium borohydride. Both lithium aluminium hydride and sodium hydride are reducing agents but LAH is more stronger.
Complete step by step solution:
- It is known that Lithium Aluminium Hydride (LAH) is a strong, unselective reducing agent for polar double bonds and is a source of hydride ions.
- LAH will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols. Also, it reduces amides and nitriles to amines.
- Sodium borohydride is also used as a reducing agent in organic chemistry. It is commonly used to reduce ketones by donating hydride ions. This compound can be used in aqueous solution because it is not that reactive compared to LAH.
- Now, coming to the question, the compound which is reacted with both sodium borohydride and LAH is 3-Phenylprop-2-enal and its common name is cinnamaldehyde.
- We know that alkene gives electrophilic addition reaction so it will not react with sodium borohydride so sodium borohydride will only reduce the carbonyl group into corresponding alcohol. The reaction can be given as under.
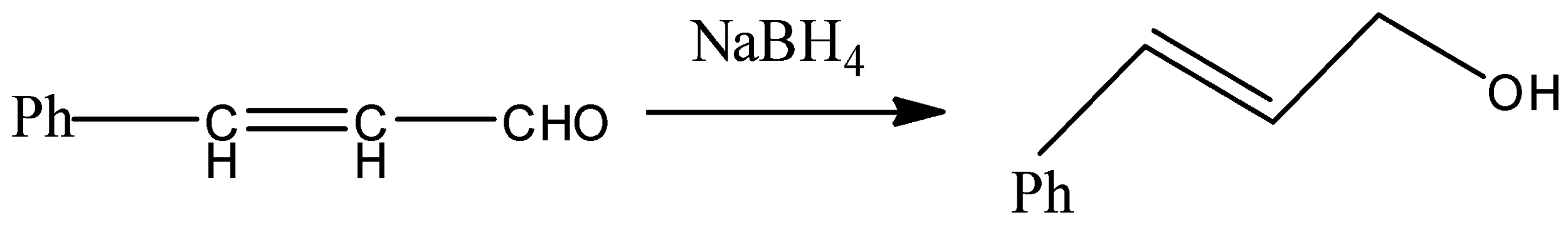
But since, the given compound is alpha-beta unsaturated aldehyde so there is conjugation between alkene and carbonyl, so with LAH carbonyl and alkenes also get reduced. That means we will get an alkene functional group converted into alkane as well.
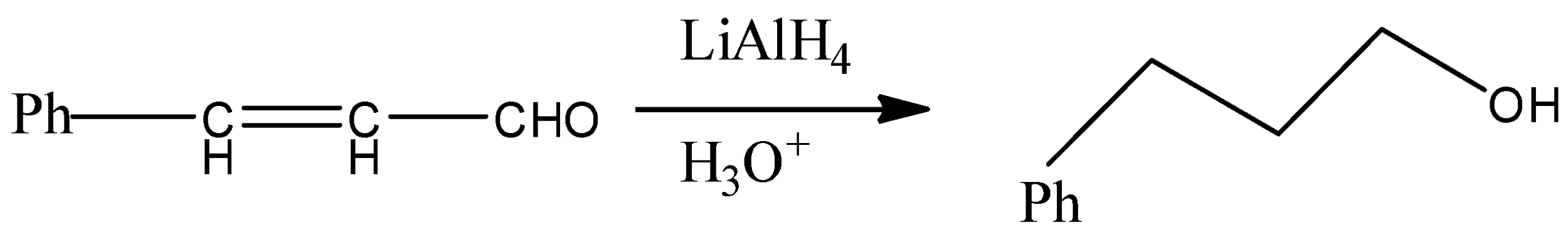
Therefore, from above we can conclude that option (D) is the correct option to the given question.
Note:
It should be remembered that in order to reduce aldehyde or ketone selectively without reducing carbon-carbon double bond in the same molecule, a reducing agent hydroborane known as 9-BBN is also used. Also, you should remember that catalytic hydrogenation can reduce both the double bond and carbonyl group simultaneously.
Complete step by step solution:
- It is known that Lithium Aluminium Hydride (LAH) is a strong, unselective reducing agent for polar double bonds and is a source of hydride ions.
- LAH will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols. Also, it reduces amides and nitriles to amines.
- Sodium borohydride is also used as a reducing agent in organic chemistry. It is commonly used to reduce ketones by donating hydride ions. This compound can be used in aqueous solution because it is not that reactive compared to LAH.
- Now, coming to the question, the compound which is reacted with both sodium borohydride and LAH is 3-Phenylprop-2-enal and its common name is cinnamaldehyde.
- We know that alkene gives electrophilic addition reaction so it will not react with sodium borohydride so sodium borohydride will only reduce the carbonyl group into corresponding alcohol. The reaction can be given as under.
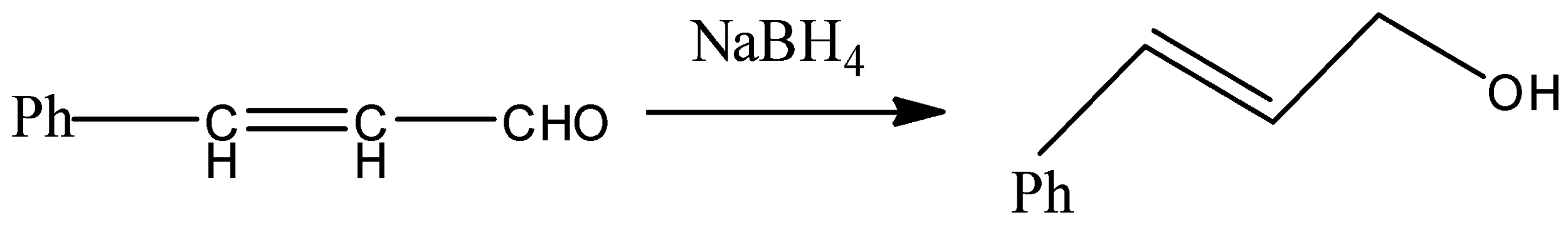
But since, the given compound is alpha-beta unsaturated aldehyde so there is conjugation between alkene and carbonyl, so with LAH carbonyl and alkenes also get reduced. That means we will get an alkene functional group converted into alkane as well.

Therefore, from above we can conclude that option (D) is the correct option to the given question.
Note:
It should be remembered that in order to reduce aldehyde or ketone selectively without reducing carbon-carbon double bond in the same molecule, a reducing agent hydroborane known as 9-BBN is also used. Also, you should remember that catalytic hydrogenation can reduce both the double bond and carbonyl group simultaneously.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




