What is the electron sea model?
(a) The model of metallic bonding where electrons float free in a sea of electrons around metal atoms.
(b) Where electrons float free in a sea of salt water.
(c) The model of metallic bonding where electrons are fixed in place in a sea of metal atoms.
(d) A model depicting the different bonds that electrons can make.
(e) The model of metallic bonding where protons float free in a sea of electrons around metal atoms.
Answer
559.6k+ views
Hint: We know most metals have low electronegativity hence the electrons are not held very tightly by the metal atoms. Therefore, we can imagine them to be moving on the surface of the metal free from each other. This forms the basis of the Electron Sea Model.
Complete step by step solution:
We can see in metallic bonding there are no atoms with higher electronegativities for the electron density to be transferred to. This could mean that in metallic bonding for the metal atom to become more stable it should release its electron density but without transferring the electrons to another atom. Hence we can say the electrons are now free to move between atoms without being bonded to any particular atom.
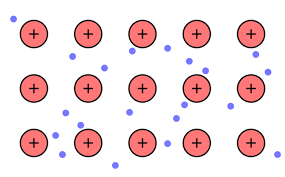
Thus, we can conclude that Option (A) the model of metallic bonding where electrons float free in a sea of electrons around metal atoms is the correct answer.
Additional information:
The free electrons on the surface are the sea of electrons. From this model of freely moving electrons we can find the explanation of the properties of electric conductivity, malleability, luster, and heat conductivity in metals. It also helps scientists to picture the behavior of electrons in metallic bonding.
Note: This model established by Dirac has some drawbacks too. There are certain characteristics that we cannot explain by this model, such as the formation of certain alloys between metals with specific compositions or the stability of collective metal bonds, among others but these are easily explained by the Quantum model.
Complete step by step solution:
We can see in metallic bonding there are no atoms with higher electronegativities for the electron density to be transferred to. This could mean that in metallic bonding for the metal atom to become more stable it should release its electron density but without transferring the electrons to another atom. Hence we can say the electrons are now free to move between atoms without being bonded to any particular atom.

Thus, we can conclude that Option (A) the model of metallic bonding where electrons float free in a sea of electrons around metal atoms is the correct answer.
Additional information:
The free electrons on the surface are the sea of electrons. From this model of freely moving electrons we can find the explanation of the properties of electric conductivity, malleability, luster, and heat conductivity in metals. It also helps scientists to picture the behavior of electrons in metallic bonding.
Note: This model established by Dirac has some drawbacks too. There are certain characteristics that we cannot explain by this model, such as the formation of certain alloys between metals with specific compositions or the stability of collective metal bonds, among others but these are easily explained by the Quantum model.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




