$d{{z}^{2}}$ orbital has :
(A) A lobe along the z-axis and a ring along xy-plane
(B) A lobe along the z-axis and a lobe along xy-plane
(C) A lobe along the z-axis and a ring along yz-plane
(D) A lobe and ring along the z-axis
Answer
265.8k+ views
Hint: Every orbital has a different shape and different lobes. The shape is decided by the azimuthal quantum number which is denoted as ‘l’. $d{{z}^{2}}$orbital is one of the 5 orbitals of d subshell which has a little different structure from ${{p}_{z}}$.
Complete step by step solution:
-There are 4 quantum numbers n, l, m and s. The principal quantum number (n) tells us about the shells of the atom. The azimuthal quantum number (l) tells us about the subshells of the atom. The magnetic quantum number (m) tells us about the orbitals of the atom and the spin quantum number (s) tells us about the orientation of the electrons in the orbital.
-Third quantum number is the magnetic quantum number and is denoted by m. It gives us the exact orbitals. Its total value can be given as ${{n}^{2}}$ or as (2l+1) since it lies in the range (-l to +l).
So, the d-subshell comes into picture when the value of n=3 and there are 5 orbitals for d-subshell since the value of l will be 2 for it. These orbitals are ${{d}_{xy}}, {{d}_{yz}}, {{d}_{xz}}, {{d}_{{{x}^{2}}-{{y}^{2}}, }}{{d}_{{{z}^{2}}}}$
-d-orbitals can be formed in 2 ways: between the axis and along the axis. $d{{z}^{2}}$ orbital is formed along the axis of the yz-plane and xz-plane. It has maximum electron density along the z-axis and is an axial orbital. The electron density of $d{{z}^{2}}$ orbital is negligible in the xy-plane and has the same phase in the opposite directions.
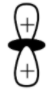
This is the shape of $d{{z}^{2}}$ orbital
So the correct option is A. A lobe along the z-axis and a ring along xy-plane.
Note: $d{{z}^{2}}$ orbital is similar to the ${{p}_{z}}$ orbital but is not the same. The probability of finding an electron is zero along the xy-plane for ${{p}_{z}}$orbital but this is not the case with the $d{{z}^{2}}$ orbital. Probability is not completely zero in it. Also, $d{{z}^{2}}$ orbital has the same phase in the opposite direction while ${{p}_{z}}$ orbital has a different phase in different directions.
Complete step by step solution:
-There are 4 quantum numbers n, l, m and s. The principal quantum number (n) tells us about the shells of the atom. The azimuthal quantum number (l) tells us about the subshells of the atom. The magnetic quantum number (m) tells us about the orbitals of the atom and the spin quantum number (s) tells us about the orientation of the electrons in the orbital.
-Third quantum number is the magnetic quantum number and is denoted by m. It gives us the exact orbitals. Its total value can be given as ${{n}^{2}}$ or as (2l+1) since it lies in the range (-l to +l).
So, the d-subshell comes into picture when the value of n=3 and there are 5 orbitals for d-subshell since the value of l will be 2 for it. These orbitals are ${{d}_{xy}}, {{d}_{yz}}, {{d}_{xz}}, {{d}_{{{x}^{2}}-{{y}^{2}}, }}{{d}_{{{z}^{2}}}}$
-d-orbitals can be formed in 2 ways: between the axis and along the axis. $d{{z}^{2}}$ orbital is formed along the axis of the yz-plane and xz-plane. It has maximum electron density along the z-axis and is an axial orbital. The electron density of $d{{z}^{2}}$ orbital is negligible in the xy-plane and has the same phase in the opposite directions.

This is the shape of $d{{z}^{2}}$ orbital
So the correct option is A. A lobe along the z-axis and a ring along xy-plane.
Note: $d{{z}^{2}}$ orbital is similar to the ${{p}_{z}}$ orbital but is not the same. The probability of finding an electron is zero along the xy-plane for ${{p}_{z}}$orbital but this is not the case with the $d{{z}^{2}}$ orbital. Probability is not completely zero in it. Also, $d{{z}^{2}}$ orbital has the same phase in the opposite direction while ${{p}_{z}}$ orbital has a different phase in different directions.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




