Distillation of phenol with zinc-dust gives:
(A) Benzene
(B) Diphenyl - zinc
(C) Diphenyl Ether
(D) none of the above
Answer
548.1k+ views
The product was discovered by Kekule. It has the molecular formula \[{{C}_{6}}{{H}_{6}}\].
It is an aromatic compound as it obeys Huckel’s rule of \[(4n+2)\pi \]electrons. It is a very stable compound due to resonance.
Complete step by step answer:
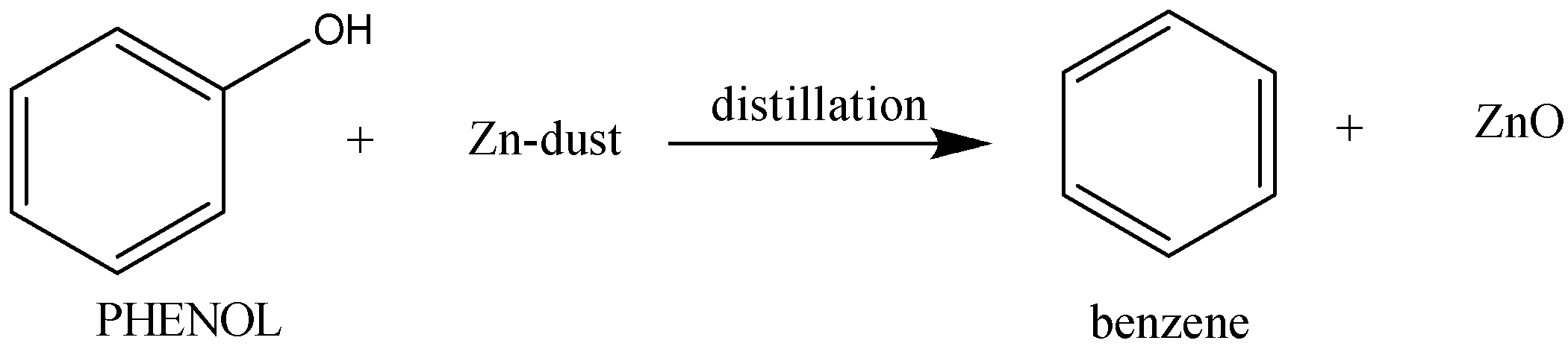
Distillation of phenol with zinc-dust gives benzene and ZnO as a side-product.
The reaction proceeds as follows:
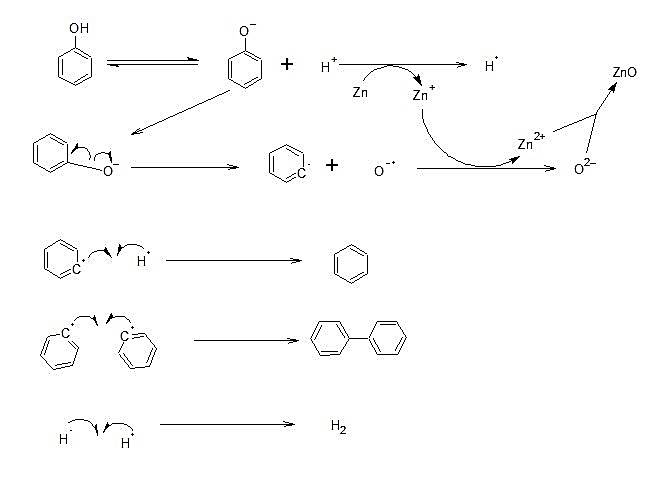
 Detailed Mechanism:
Detailed Mechanism:
- Zn is in the +2 oxidation state.
- The phenol gets converted into phenoxide ion and the released proton accepts an electron from Zn forming hydrogen radical. Due to the heating, there is homolytic fission of C of the phenyl ring and\[{{O}^{-}}\].
- Then\[{{O}^{-}}\]forms an electron from Zn and forms an oxide ion. In this way, zinc forms zinc oxide, and the phenyl radical produced forms a bond with hydrogen radical to form Benzene and bi-phenyl.
So, the correct option is A.
Note: The yield of this reaction used for benzene formation is lower.
Homolytic fission is the equal splitting of a pair of electrons between two separated atoms.
It is a reduction reaction of phenol to benzene by zinc dust. So, zinc is a reducing agent.
Benzene formed is volatile in nature. It is separated by fractional distillation.
It is an aromatic compound as it obeys Huckel’s rule of \[(4n+2)\pi \]electrons. It is a very stable compound due to resonance.
Complete step by step answer:
Distillation of phenol with zinc-dust gives benzene and ZnO as a side-product.
The reaction proceeds as follows:
 Detailed Mechanism:
Detailed Mechanism:- Zn is in the +2 oxidation state.
- The phenol gets converted into phenoxide ion and the released proton accepts an electron from Zn forming hydrogen radical. Due to the heating, there is homolytic fission of C of the phenyl ring and\[{{O}^{-}}\].
- Then\[{{O}^{-}}\]forms an electron from Zn and forms an oxide ion. In this way, zinc forms zinc oxide, and the phenyl radical produced forms a bond with hydrogen radical to form Benzene and bi-phenyl.

So, the correct option is A.
Note: The yield of this reaction used for benzene formation is lower.
Homolytic fission is the equal splitting of a pair of electrons between two separated atoms.
It is a reduction reaction of phenol to benzene by zinc dust. So, zinc is a reducing agent.
Benzene formed is volatile in nature. It is separated by fractional distillation.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




