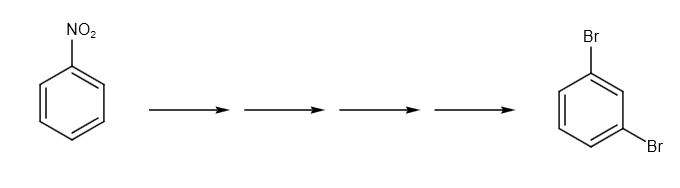
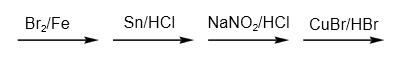
What is the correct sequence of reagent used for converting nitrobenzene into m-dinitrobenzene?
A.
B.
C.
D.
Answer
254.4k+ views
Hint: The aromatic chemical known as nitrobenzene contains a nitro group inside of a benzene ring. The Nitro group is meta-directing. Hence, any forward reaction will be taking place at the meta position of the ring.
Complete Step by Step Solution:
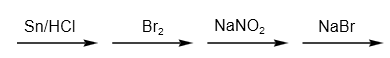
Firstly, we have to do bromination of the nitrobenzene. The reagent used for the reaction is \[B{r_2}/Fe\] reagent. Here, electrophilic substitution takes place. The reaction is as follows:
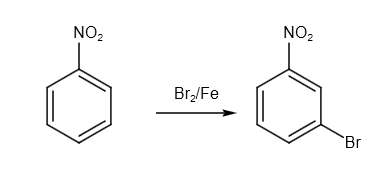
Image: Bromination of nitrobenzene
After that, we have to convert \[N{O_2}\] group to \[N{H_2}\] group. So, we can use \[Sn/HCl\] reagent. Tin (IV) chloride is created when tin interacts with HCl. Consequently, we may state that \[{H^ + }\] ions are released as a result of the aromatic compound's nitro functional group reaction. The reaction is as follows:
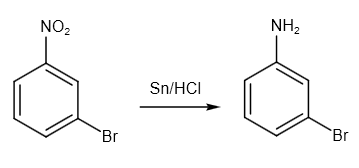
Image: Reduction by tin and HCl reagent
After that, we have to convert it into diazonium salt. The compound reacts with sodium nitrite and HCl in cold conditions to form diazonium salt. The reaction is as follows:
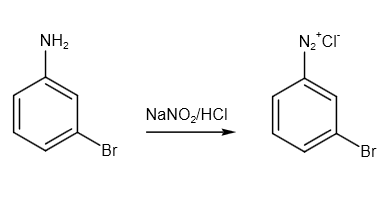
Image: Formation of diazonium salt
Finally, we have to use the Sandmeyer reaction to get the desired product dibromo benzene. A common substitution reaction is the Sandmeyer reaction, which is used to produce aryl halides from aryl diazonium ions. Catalysts for this process include copper salts like chloride, bromide, or iodide ions. The reaction is as follows:
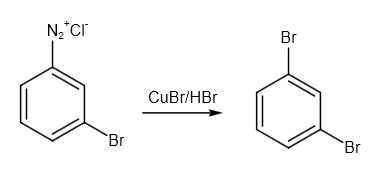
Image: Sandmeyer reaction
The complete reaction is written as follows:
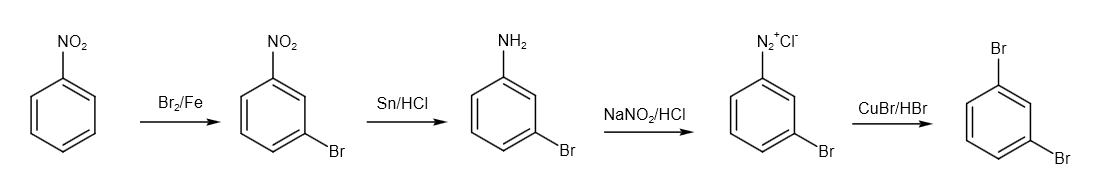
Image: Conversion of m-dinitrobenzene to dibromobenzene
As a result, the correct order of reagents used is option D.
Note: The two-step mechanism reaction is the Sandmeyer reaction. The free radical mechanism is used in the Sandmeyer reaction. The halogen linked to the copper enters the benzene ring during this process.
Complete Step by Step Solution:
Firstly, we have to do bromination of the nitrobenzene. The reagent used for the reaction is \[B{r_2}/Fe\] reagent. Here, electrophilic substitution takes place. The reaction is as follows:

Image: Bromination of nitrobenzene
After that, we have to convert \[N{O_2}\] group to \[N{H_2}\] group. So, we can use \[Sn/HCl\] reagent. Tin (IV) chloride is created when tin interacts with HCl. Consequently, we may state that \[{H^ + }\] ions are released as a result of the aromatic compound's nitro functional group reaction. The reaction is as follows:

Image: Reduction by tin and HCl reagent
After that, we have to convert it into diazonium salt. The compound reacts with sodium nitrite and HCl in cold conditions to form diazonium salt. The reaction is as follows:

Image: Formation of diazonium salt
Finally, we have to use the Sandmeyer reaction to get the desired product dibromo benzene. A common substitution reaction is the Sandmeyer reaction, which is used to produce aryl halides from aryl diazonium ions. Catalysts for this process include copper salts like chloride, bromide, or iodide ions. The reaction is as follows:

Image: Sandmeyer reaction
The complete reaction is written as follows:

Image: Conversion of m-dinitrobenzene to dibromobenzene
As a result, the correct order of reagents used is option D.
Note: The two-step mechanism reaction is the Sandmeyer reaction. The free radical mechanism is used in the Sandmeyer reaction. The halogen linked to the copper enters the benzene ring during this process.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




