Compounds containing both amino and COOH groups are known as
A. Diamines
B. unknown
C. Amino acids
D. Enzymes
Answer
249k+ views
Hint: The amino group is a functional group which is present in an amine. COOH is the functional group which is present in a carboxylic acid compound.
Complete Step by Step Solution:
Functional groups are the groups that present chemical compounds which show their different chemical properties. Different functional groups show different properties.
Amino acids are organic compounds which combine together to form a protein. Therefore, amino acids are referred to as the building block of proteins.
An amino group is a functional group with the general formula \[R - N{H_2}\]where R is the alkyl group.
The Carboxyl group is a functional group with the general formula R-COOH where R is the alkyl group.
Amino acid is an organic molecule which contains both the amino functional group and carboxyl functional group attached to the central carbon atom. The two bonds of carbon are attached to an alkyl group (R) and a hydrogen atom.
The general structure of amino acids is given below.
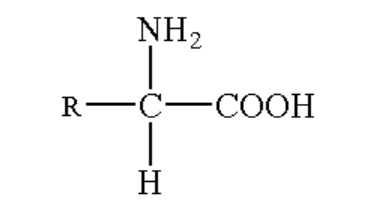
Image: General formula of amino acid.
Thus, the compounds containing both amino and COOH groups are known as amino acids.
Therefore, option C is correct.
Note: There are different amino acids which differ from each other on the basis of the alkyl group attached. There is a total of 20 amino acids which occur naturally among which there are a total of 9 essential amino acids i.e, histidine, leucine, lycine, isoleucine, methionine, valine, phenylalanine, threonine, and tryptophan.
Complete Step by Step Solution:
Functional groups are the groups that present chemical compounds which show their different chemical properties. Different functional groups show different properties.
Amino acids are organic compounds which combine together to form a protein. Therefore, amino acids are referred to as the building block of proteins.
An amino group is a functional group with the general formula \[R - N{H_2}\]where R is the alkyl group.
The Carboxyl group is a functional group with the general formula R-COOH where R is the alkyl group.
Amino acid is an organic molecule which contains both the amino functional group and carboxyl functional group attached to the central carbon atom. The two bonds of carbon are attached to an alkyl group (R) and a hydrogen atom.
The general structure of amino acids is given below.

Image: General formula of amino acid.
Thus, the compounds containing both amino and COOH groups are known as amino acids.
Therefore, option C is correct.
Note: There are different amino acids which differ from each other on the basis of the alkyl group attached. There is a total of 20 amino acids which occur naturally among which there are a total of 9 essential amino acids i.e, histidine, leucine, lycine, isoleucine, methionine, valine, phenylalanine, threonine, and tryptophan.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

Understanding the Angle of Deviation in a Prism

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




