What is the colour of methyl orange in acidic and basic medium respectively?
(A) Yellow, Red
(B) Red, Yellow
(C) Colourless, Red
(D) Colourless, Yellow
Answer
268.2k+ views
Hint: Methyl orange is an organic compound which is used as a laboratory agent. It is mostly used as a pH indicator frequently used in titrations due to its distinct colour and variance at different pH values.
Complete step by step answer: Methyl orange is a chemical compound having chemical formula ${C}_{14}{H}_{14}{N}_{3}Na{O}_{3}S$.
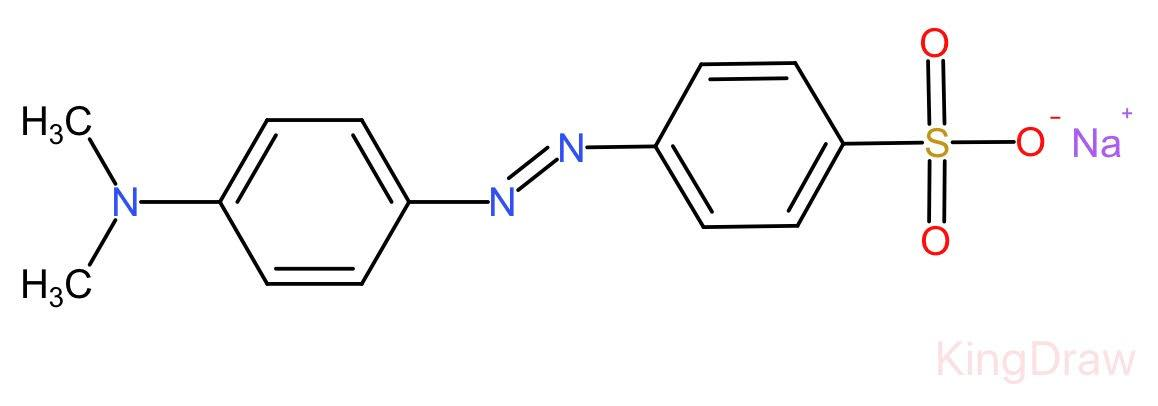
Unlike a universal indicator, it does not show a full spectrum of colour change but does have a sharp endpoint.
- In a solution that is becoming less acidic in nature, methyl orange tends to change its colour from red to orange and finally to yellow. This entire colour change occurs in acidic conditions.
- Methyl orange shows a red colour when the pH of the solution is less than or equal to 3.1 and shows a yellow colour when the pH is above or equal to 4.4.
- As the changes occur in the $p{k}_{a}$ of a mid-strength acid, methyl orange is usually used for the titration of acids.
Therefore, in the acidic medium, it is red in colour and in the basic medium, it is yellow in colour.
Hence, option (b) is the correct answer.
Note: Methyl orange is insoluble in diethyl ether, but its solubility in water is 0.5 g/100mL (20 degree Celsius). It is an orange solid in appearance and direct contact with methyl orange should be avoided.
Complete step by step answer: Methyl orange is a chemical compound having chemical formula ${C}_{14}{H}_{14}{N}_{3}Na{O}_{3}S$.

Unlike a universal indicator, it does not show a full spectrum of colour change but does have a sharp endpoint.
- In a solution that is becoming less acidic in nature, methyl orange tends to change its colour from red to orange and finally to yellow. This entire colour change occurs in acidic conditions.
- Methyl orange shows a red colour when the pH of the solution is less than or equal to 3.1 and shows a yellow colour when the pH is above or equal to 4.4.
- As the changes occur in the $p{k}_{a}$ of a mid-strength acid, methyl orange is usually used for the titration of acids.
Therefore, in the acidic medium, it is red in colour and in the basic medium, it is yellow in colour.
Hence, option (b) is the correct answer.
Note: Methyl orange is insoluble in diethyl ether, but its solubility in water is 0.5 g/100mL (20 degree Celsius). It is an orange solid in appearance and direct contact with methyl orange should be avoided.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




