Aromatic primary amine when treated with cold \[HN{O_2}\] gives
(A) Benzyl Alcohol
(B) Nitro Benzene
(C) Benzene
(D) Diazonium salt
Answer
267.9k+ views
Hint: An amine is often a functional group with a lone pair on the nitrogen atom. Amines structurally mimic ammonia, where nitrogen can link with up to three hydrogen atoms. It is also defined by a number of carbon connectivity-based features.
Complete Step by Step Solution:
In order to know that when an alkyl or aryl group replaces one of the hydrogen atoms in the ammonia molecule, the presence of the primary amines can be determined. For instance: Methylamine \[{\text{ }}C{H_3}N{H_2}\]and Aniline \[{C_6}{H_5}N{H_2}\].
For the simple removal of a water molecule, nitrous acid \[(HN{O_2})\] is represented by the formula \[HO - N = O\]. As a result, a water molecule is created when the secondary amine's hydrogen ion joins with nitrous acid's \[O{H^ - }\]ion.
The reaction process that occurs when amine reacts with cold \[HN{O_2}\] will now be discussed below:
Using nitrous acid \[(HN{O_2})\]reagent to convert amine to diazonium ion,
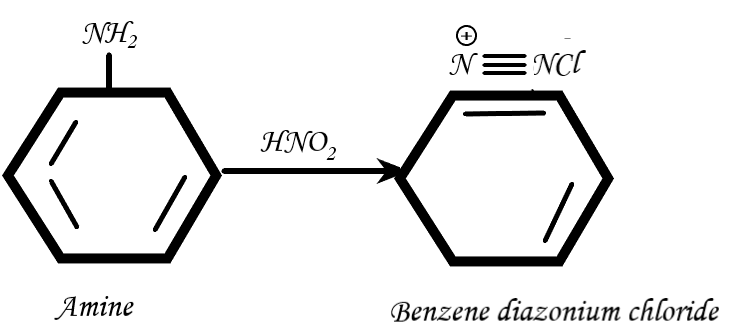
A primary aromatic amines react with cold \[HN{O_2}\]and dissolved in dilute \[HCl\]at \[0 - 5^\circ C\]will produced diazonium salt. The nitrogen gas will emerge when the cold salt is heated to the room temperature.
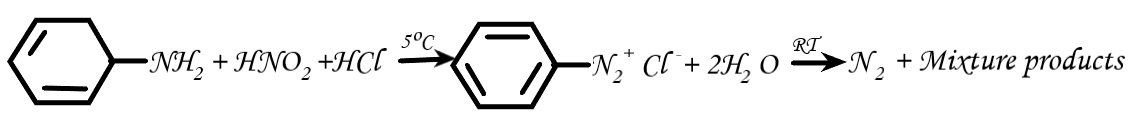
Therefore, the correct option is: (D) Diazonium salt.
Note: It is important to remember that the carbylamine reaction is utilised to find primary amines. Isocyanide, a substance that is produced and has a strong odour, is the end result. This reaction is not produced by secondary or tertiary amines. The reaction is as following: \[RN{H_2} + CHC{l_3} + 3KOH \to RNC + 3KCl + 3{H_2}O\]
Complete Step by Step Solution:
In order to know that when an alkyl or aryl group replaces one of the hydrogen atoms in the ammonia molecule, the presence of the primary amines can be determined. For instance: Methylamine \[{\text{ }}C{H_3}N{H_2}\]and Aniline \[{C_6}{H_5}N{H_2}\].
For the simple removal of a water molecule, nitrous acid \[(HN{O_2})\] is represented by the formula \[HO - N = O\]. As a result, a water molecule is created when the secondary amine's hydrogen ion joins with nitrous acid's \[O{H^ - }\]ion.
The reaction process that occurs when amine reacts with cold \[HN{O_2}\] will now be discussed below:
Using nitrous acid \[(HN{O_2})\]reagent to convert amine to diazonium ion,

A primary aromatic amines react with cold \[HN{O_2}\]and dissolved in dilute \[HCl\]at \[0 - 5^\circ C\]will produced diazonium salt. The nitrogen gas will emerge when the cold salt is heated to the room temperature.

Therefore, the correct option is: (D) Diazonium salt.
Note: It is important to remember that the carbylamine reaction is utilised to find primary amines. Isocyanide, a substance that is produced and has a strong odour, is the end result. This reaction is not produced by secondary or tertiary amines. The reaction is as following: \[RN{H_2} + CHC{l_3} + 3KOH \to RNC + 3KCl + 3{H_2}O\]
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




